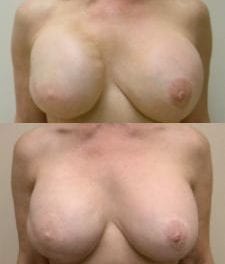
Careful preparation leads to a surgical plan that ensures a successful breast-augmentation revision
 Unlike the mediocre result of a facelift, the mediocre result of a breast augmentation can be covered by most attire. Only the patient and the surgeon need be aware that results fell short of what may have been expected. Once unveiled in the examining room, however, the cause of a poor result in breast augmentation can often be easily determined.
Unlike the mediocre result of a facelift, the mediocre result of a breast augmentation can be covered by most attire. Only the patient and the surgeon need be aware that results fell short of what may have been expected. Once unveiled in the examining room, however, the cause of a poor result in breast augmentation can often be easily determined.
Revisional breast-augmentation surgery is rewarding because patients are usually appreciative and the results are good—and are usually long-lasting.
Indications
Revision candidates fall into the five groups summarized in the following paragraphs:
1) The first group of patients who can be helped by revision are those with grade III and grade IV capsular contractures that cause discomfort during normal activities. It is useful to try to determine—from the patient’s history, previous operative notes, and postoperative course—the possible cause of the contracture: whether it arose from previous complications such as hematoma or infection, or from ruptured silicone material in the capsule. When there is no apparent cause, you must consider the patient a “capsule former” who might heal similarly after revision.
2) The second patient group has mechanical failure of the implant. Early deflation of a saline implant can be caused by shell wear from overfilling or underfilling (more than 10% outside the manufacturer’s recommended range). This has been my personal experience, and it has also been reported by implant companies.1
Although a ruptured silicone implant can present asymptomatically, it frequently manifests itself as a breast lump or a clinically significant capsular contracture that has been confirmed mammographically. Patients sometimes tell me that their previous surgeon did not recommend removing a ruptured silicone implant, but if it were my silicone implant, I would want it removed and replaced. When replacement is not medically feasible, removal is a good compromise.
3) The third group of patients have gross distortion from malpositioning of one or both implants. Malpositioning causes measurable asymmetry between the landmarks of the breasts. Slight asymmetry can be tolerated by most patients, although it will not build the breast surgeon’s reputation. Asymmetry detectable in a bathing suit, however, is a nuisance, and patients will eventually seek help.
4) Patients in the fourth group believe that they are underaugmented or overaugmented. A treatise could be written about the reasons for and against complying with a request for a change in implant size, but the core argument is whether the current implant size distorts or improves the aesthetic of the whole-body habitus. Does the current implant meet the golden proportion rule of a bust–waist–hip ratio of 1:0.7:1?
A plastic surgeon can feel confident in recommending any size implant that approximates this proportion and should feel hesitant about a size that ignores this guidance. If the patient is adamant about ignoring the surgeon’s advice, a simple discussion about the risks of poor healing, bottoming out, and stretch relaxation of tissues is necessary and should be supported with documentation. Whether to perform the surgery is, ultimately, the surgeon’s decision.
5) The fifth group of patients have uncorrected breast ptosis, whether primary (probably preoperative, but not addressed by the previous surgeon) or secondary (due to the current augmentation and its anatomical consequences). In my experience, most patients prefer acceptable scars on the breasts—if they confer a better breast shape, good enough to wear a tank top with no bra—to ptotic breasts with no scars. The obvious exception is the patient with a history of poor scar healing, the cause of which needs to be elucidated and corrected, if possible.
Contraindications
Assuming that the prospective patient is an acceptable anesthesia risk, there are two main medical contraindications to proceeding with revisional breast surgery. Previously undiagnosed pathology found during breast examination, mammography, or magnetic resonance imaging scanning needs to be diagnosed prior to surgery. In my practice, every patient older than 25 years and every patient who has been pregnant undergoes a preoperative mammogram.
If the patient has a mass or calcification, a breast-specialized general surgeon performs the diagnostic biopsy and I wait at least 8 weeks to perform surgery. I also consider a patient who has had multiple procedures and who has tight, scarred tissues a poor immediate revision candidate but a possible implant-removal candidate. I will ask the patient to consider living without implants for 6 to 12 months so that the tissues can shrink and develop a more normal vascularity, possibly laying the groundwork for a better future result.
I tell these patients that I myself developed a periprosthetic infection and chose to live without my implants for 1½ years. The experience was not that bad, and it gave me an appreciation for what we do to patients for the sake of aesthetics. My story makes them realize that there are always alternatives as long as the patient stays healthy.
Choosing the Procedure
Historically, the procedures used to revise an undesirable breast augmentation have varied, and each solves a different anatomical problem. Deflation of a saline implant requires a limited capsulotomy and gives the surgeon the option of increasing or decreasing the size of the implant or changing its position.
Malpositioning of an implant requires a capsulotomy and then a capsulorrhaphy.1 When capsular contracture accompanies a deflated saline implant, a more complete capsulotomy in radial and perimeter locations opens up a contracted bed and allows better positioning of the implant.
With a ruptured silicone implant, especially in a scarred bed, partial or total capsulectomy with conversion to a virgin plane has been recommended.1 Some authors prefer to dissect only one side of the capsule and then move the implant to a virgin pocket.2 Breast ptosis of grade III or IV severity, previously uncorrected, requires a periareolar, keyhole, or standard mastopexy.1
Preoperative Factors
A precise preoperative evaluation of the patient and her problem allows the surgeon to formulate a successful game plan. A problem-focused medical history must investigate any occurrence of breast cancer in the family and any existing pathology suggested by examination or mammography.
Bleeding tendencies cause hematoma or increased scarring around the new pocket, so any history of smoking or the use of nonsteroidal anti-inflammatory drugs, vitamin E, fish oil, or herbal remedies needs to be elicited; the patient must discontinue any use of agents that interfere with coagulation. The patient’s chief complaint and the anatomical cause for it should be determined.
I use the measurement system developed by John B. Tebbetts, MD, in 19903 and revised in 1994.4 Distance from sternal notch to nipple, base breast width (lateral sternal border to anterior axillary line), distance from areola to inframammary fold, and discrepancies in the level of the inframammary folds are useful measurements for documenting existing anatomy and for setting goals.
Very often, chest-wall asymmetries that were not recognized by the original surgeon contribute to implant-positioning problems and perceived volume asymmetries. Planning the new surgery for masking chest-wall asymmetry will almost certainly ensure success.
The original surgical notes should provide data about the existing implants’ sizes and types. Changes in the size and type must be considered if they might help accomplish the new goals. The exact surgical maneuvers—whether subpectoral conversion, capsulorrhaphy, or mastopexy—need to be defined, along with how the new or old implant—smooth or textured, saline or gel—will affect the outcome.
Technical Considerations
All patients are premedicated using 200 mg of celecoxib, twice daily, starting 1 day before surgery. A scopolamine patch (2 mg) is applied the night before surgery and removed on the morning of the first postoperative day.
The patient is marked in the preoperative holding bay while sitting up straight on an examination bench with shoulders level and rib cage expanded; a retractable tape measure and black marker are used. The sternal midline, the lateral sternal border, the upper and lateral limits of the dissection, the planned new inframammary crease, the limited 3-cm inframammary-fold incision, and the new planned location (if needed) for the nipple–areolar complex are marked thickly.
All of my surgeries are done under general anesthesia. All patients are infiltrated at the incision site, and then at the prepectoral level, with 280 mL of 0.125% lidocaine (250 mL normal saline solution with one ampule of 1:1000 epinephrine and 30 mL of 1% lidocaine hydrochloride with 1:100,000 epinephrine added).
Infiltration is started using a 30-mL syringe attached to a 21-gauge spinal needle, then continued with an 18-gauge spinal needle. Complete infiltration into the medial costal region of the pectoral muscle and lateral intersection with the serratus anterior are used to control bleeding during the scissor and electrosurgical dissection.
All patients are drained through a stab incision at the low anterior axillary line using a 7-mm bulb-reservoir suction drain until the 24-hour accumulation is less than 30 mL per side. Preoperative cephazolin (1 g, delivered intravenously) is continued as 500 mg of cefadroxil, twice daily, until the drains are removed, usually 4 to 6 days after surgery.
Postoperative dressings include adhesive strips on incisions, 4- x 4-inch gauze pads, and closed-cell foam stretch tape. When the patient is seen on the first postoperative visit, 2 days after surgery, these dressings are exchanged for a surgical bra with a front closure, which is kept on for 2 weeks.
All patients are given instructions in implant massage starting 1 week after surgery. They are told to do these exercises for 10 seconds for each breast, five times per day, for 1 year.
Clinical Cases
Case 1. A 35-year-old hand therapist presented with the complaint that her implants were falling down her chest and were too far apart. She was very muscular, and when she flexed her pectoralis major muscles they pushed down the implants.
On examination, I found that her inframammary fold had been completely disrupted by inferior overdissection. She also had an uncorrected narrow-based breast deformity, and her 300-mL implants had been filled to 450 mL. Her skin was of poor quality because of stretch marks and previous weight loss.
 My diagnosis was loss of anatomical inframammary fold, grade III breast ptosis, and underaugmentation by use of an implant with too narrow a base. I proposed a two-stage operation. It was difficult to get the patient to agree, but she finally did so after 9 months.
My diagnosis was loss of anatomical inframammary fold, grade III breast ptosis, and underaugmentation by use of an implant with too narrow a base. I proposed a two-stage operation. It was difficult to get the patient to agree, but she finally did so after 9 months.
In the first stage, implant removal and inferior capsulectomies and capsulorraphies with 2-0 coated polybutylate nonabsorbable suture material re-established the inframammary folds. The absence of implants allowed the inframammary fold to heal without tension and permitted the skin to shrink to its natural limits.
In the second stage, the upper submuscular pole was redissected generously to allow the bulk of the implant to have a submuscular placement. The patient received 450-mL implants that were filled to 460 mL on the right and 430 mL on the left. Concentric mastopexies with a short inferior scar lifted the nipple–areolar complex to the apex of the new breast mound. The patient is much happier with her breasts, but wishes that there was not the slightest parenchyma-implant step-off in the left lower pole (Figure 1, page 32).
Case 2. A 38-year-old homemaker had undergone breast augmentation using 275-mL implants 4 years earlier. She immediately noticed increased breast asymmetry, but was told by her surgeon that nothing could be done about it. She presented with the complaint that her left breast would fall out of her bra into her axilla.
On examination, the distance from her sternal notch to nipple was noted as 20.5 cm on the right and 23 cm on the left. The left implant was lying very laterally and low on the chest wall.
I diagnosed asymmetry due to malpositioning of the left breast implant through overdissection of the left lateral gutter, along with glandular ptosis, grade III, of the left nipple–areolar complex. I have seen two other patients with the same problem, and I presume that a right-handed surgeon may unwittingly overdissect the left lateral gutter using a nonendoscopic transaxillary subpectoral approach.
 This patient’s surgery involved redissection of only the upper and medial subpectoral pockets bilaterally, a left capsulorrhaphy with 2-0 coated polybutylate nonabsorbable suture material, reaugmentation using the same implants filled to 300 mL, and a left mastopexy. I find it easier to put in a few interrupted coated polybutylate nonabsorbable sutures to establish the correction and then oversew that line with a running row, starting near the axilla and working toward the incision (Figure 2, page 32).
This patient’s surgery involved redissection of only the upper and medial subpectoral pockets bilaterally, a left capsulorrhaphy with 2-0 coated polybutylate nonabsorbable suture material, reaugmentation using the same implants filled to 300 mL, and a left mastopexy. I find it easier to put in a few interrupted coated polybutylate nonabsorbable sutures to establish the correction and then oversew that line with a running row, starting near the axilla and working toward the incision (Figure 2, page 32).
Case 3. A 40-year-old hair stylist presented with an uncomfortable, unsatisfactory augmentation performed 8 years earlier. The original surgery included a subglandular augmentation using 225-mL implants filled to 350 mL.
On examination, she had very firm breasts with a distance from sternal notch to nipple of 22 cm bilaterally, a breast width of 12.5 cm bilaterally, and pseudoglandular ptosis cause by the heaviness of the implants. I diagnosed grade III capsular contracture, grade II breast ptosis, and distortion from overfilled implants.
At surgery, the anterior capsule was left intact to aid the vascularity of the nipple, and a posterior-only capsulectomy was performed, with care taken not to injure the ventral side of the pectoralis major. A subpectoral dissection was then performed, and 380-mL implants were placed. Bilateral mastopexies adjusted the skin envelope to the new breast volume and established the nipple–areolar complex on the apex of the mound. This patient loves her breasts now and has sent me many patients from her locality (Figure 3).
 A Plan and a Pearl
A Plan and a Pearl
Surgical correction of poor results in breast augmentation is made possible by correct diagnosis, formulating a surgical plan that the patient accepts, and executing that plan without complications. I have performed 905 primary breast augmentations and 188 augmentation revisions during the past 15 years of practice. I have used the analytical paradigms of Tebbetts exclusively almost since their inception.
In particular, I believe that matching the breast-base width to the base diameter of the implant will, at a minimum, produce a breast volume that is very close to optimal. I prefer textured implants because, when they are placed in a relatively scarred or virgin pocket, they stay where they are placed. This is quite an advantage in revisional surgery.
Tebbetts relayed a pearl to me that I use frequently: Just before closing the inframammary incision, put a finger under the chest-wall side of the implant and lift it up; then pull it down to the bottom of the pocket, where it should be positioned when the patient is sitting. I choose the implant style—textured or smooth—based on which implant will exert the forces on the existing muscle–parenchyma–skin envelope composite that will create the desired breast shape for that dissected pocket.
Complications
Complications include seroma, infected seroma, and (rarely) recurrence of some of the presenting deformity. Patients with suspected seroma arising after the drains have been removed are sent to a radiology facility that performs ultrasound-guided aspiration with a 22-gauge plastic catheter, minimizing the risk of implant puncture. A seroma can cause the implant to rotate, producing distortion in textured, shaped implants; but this is not a problem with round implants.
A periprosthetic infection must be dealt with immediately. I generally follow the guidelines described by Spear et al.5 Recurrence of the same deformity is rare and could be caused by suture disruption due to physical activity undertaken too early or to chest-wall deformities that allow the new implant to settle in the wrong location.
It is our role, as plastic surgeons, to help each patient to be beautiful, healthy, and whole, as much as their good health and our good surgical judgment permit. Revising mediocre breast-augmentation results is challenging and rewarding for both the patient and the surgeon.
Lori L. Cherup, MD, FACS, is a board-certified plastic surgeon and the founder and director of Radiance Plastic Surgery LLC in Bridgeville, Pa. She can be reached at [email protected].
Tiffany A. Gleason, PA-C, is a physician assistant at Radiance Plastic Surgery and the owner of Touch of Tiffany Salon and Day Spa in Greensburg, Pa.
References
1. Bostwick J III. Plastic and Reconstructive Breast Surgery. St Louis, Mo: Quality Medical Publishing; 1990:I.
2. Mladick RA. Treatment of the firm augmented breast by capsular stripping and inflatable implant exchange. Plast Reconstr Surg. 1977;60: 720–724.
3. Tebbetts JB. Dimensional Augmentation Mammoplasty Using the BioDimensional System. Santa Barbara, Calif: McGhan Medical Corp; 1994.
4. Tebbetts JB, Adams WP. Five critical decisions in breast augmentation using five measurements in 5 minutes. Plast Reconstr Surg. 2005;116: 2005–2012.
5. Spear SL, Howard MA, Boehmler JH, Ducic I, Low M, Abbruzzesse MR. The infected or exposed breast implant: Management and treatment strategies. Plast Reconstr Surg. 2004;113:1634–1644.