By Christopher Godek, MD, FACS, and Mark S. Andrew, MD
Approved by the FDA in 2010, Andrew Technologies LLC’s HydraSolve™ Lipoplasty System is a noncutting, nonthrusting, target-tissue-specific method of fat removal. It uses a novel technology called tissue liquefaction, which was borrowed from cataract surgery. This new category of lipoplasty (tissue liquefaction liposuction (TLL)) utilizes a stream of warmed (37?C to 55?C) low-pressurized (300 to 1100 psi), and pulsed (25 µL/pulse, 31 pps) saline that is emitted inside the distal end of the cannula and remains inside the cannula until aspirated.
The saline stream is not ejected into the subcutaneous space. Instead, subcutaneous fat tissue, as well as nonfat tissue, are drawn into the cannula through the aperture(s) and are impacted by the saline stream in the interior space of the cannula. The stream causes a targeted phase transition of fat tissue from a solid to a liquid, while nonfat tissue is not liquefied.
The thermal energy of the saline stream is strong enough to liquefy fat tissue but is not strong enough to liquefy nonfat tissue. Also, there is not enough thermal energy to burn any tissue. The pressure of the saline stream is also too low to cut tissue. Therefore, only the targeted fat is liquefied inside the cannula and simultaneously aspirated. The nonfat tissue is not changed and egresses back into the subcutaneous space. In addition to the lipoplasty indication, Andrew Technologies LLC recently received marketing clearance for autologous fat transfer following the HydraSolve liposuction procedure.
CASE REPORT
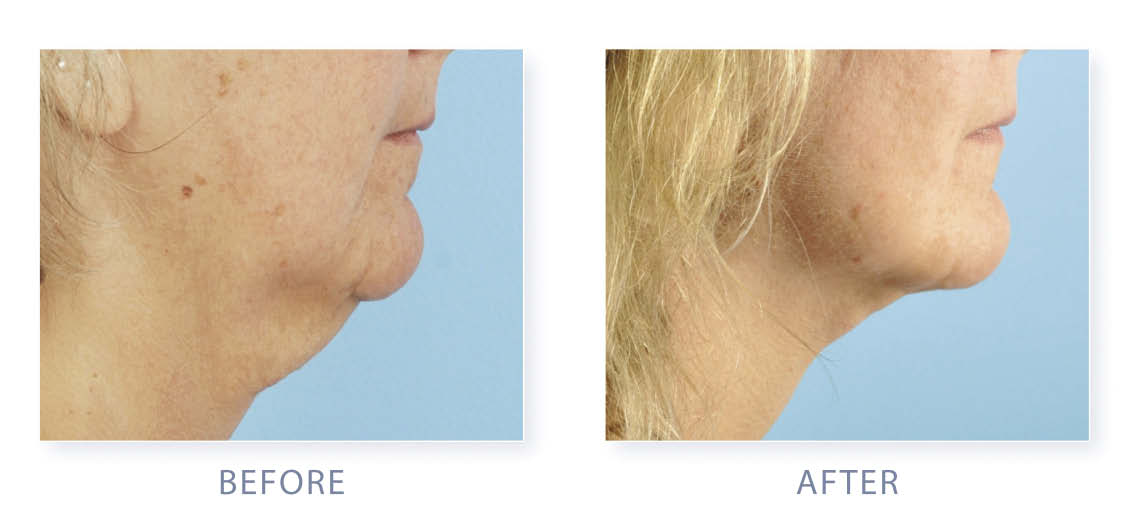
A 50-year-old Caucasian woman (weight 180 pounds, height 5 feet 8 inches) underwent a face and neck lift under general anesthesia in July 2010. She presented with lower facial laxity, neck laxity, and submental lipodystrophy. Her skin tone was fair with significant submental fat. She had firm fibrous, fatty tissue in the submental region; thick, deep, platysmal bands; with moderate mandibular glands and moderate jowl formation.
Following surgical preparation, facial areas were infiltrated with 10 mL of 0.5% lidocaine/1:100,000 epinephrine. Field infiltration was also performed with 10 mL 0.25% bupivacaine/1:200,000 epinephrine. A total of 600 mL of standard tumescent fluid was distributed to the cheeks, neck, and submental regions. A standard submuscular aponeurotic system (SMAS) facelift was performed with sideburn, retrotragal, and posterior incisions. The lateral neck was defatted with TLL (HydraSolve) via the lateral facelift incision. The fat was sculpted off the platysma muscle.
 |
|
|
A 50-year-old woman with lower facial laxity, neck laxity, and submental lipodystrophy. |
One year after facelift with TLL. |
Submental liposuction with TLL (HydraSolve; 2-mm cannula, 1 port, 900 PSI at 45?C) was performed, and 25 mL of fat was aspirated with minimal blood loss. Following hemostasis, the skin was pulled perpendicular to the nasolabial fold, excess skin was trimmed, two 7-mm flat JP drains were placed, and the incision was closed with 4-0 PDS sutures deep behind the ear, 5-0 Monocryl deep sutures in front of the ear, 6-0 Prolene sutures in front of the ear, and 5-0 Chromic sutures behind the ear. The submental closure was performed in multilayers with 5-0 Monocryl and 6-0 Prolene, and antibiotic ointment was applied. Xeroform 4x4s, 4-inch Kerlix, and 4-inch Coban were used. The patient tolerated the procedures well with no complications.
The 1-year postoperative results reveal smooth contour, diminished submental fat, and an improved neck and jaw line.
CONCLUSION
This new FDA-approved lipoplasty system utilizing TLL may be a safe, effective, and efficient mechanism for fat extraction during aesthetic body contouring procedures. TLL’s mechanism of action eliminates the need to rapidly and aggressively thrust the cannula. Therefore, the aggressive tissue shearing seen with traditional lipoplasty is not required, which may help to minimize damage to nontarget tissues. This new system allows for rapid adipose extraction with minimal surgeon fatigue. Patients recovered quickly with minimal pain, bruising, and no observed complications.
Christopher Godek, MD, FACS, is a plastic surgeon at The Personal Enhancement Center in Toms River, NJ. Mark S. Andrew, MD, is the founder of Andrew Technologies LLC. Reach them at [email protected].



