Get ready for a new crop of neurotoxin alternatives | Plastic Surgery Practice September 2014
By Wendy Lewis
 The essential role of Botulinum toxin Type A (BTX-A) in the management of facial lines and wrinkles, as well as other aesthetic and therapeutic indications, is undeniable. The neurotoxin has a high safety profile, reflected by the sparse record of side effects, as well as off-the-charts patient satisfaction due to the predictability of results, affordability, and lack of associated downtime.
The essential role of Botulinum toxin Type A (BTX-A) in the management of facial lines and wrinkles, as well as other aesthetic and therapeutic indications, is undeniable. The neurotoxin has a high safety profile, reflected by the sparse record of side effects, as well as off-the-charts patient satisfaction due to the predictability of results, affordability, and lack of associated downtime.
But there remains a subset of patients who are averse to having a foreign substance injected into their muscles, or who have yet to come around to the idea of a drug made from the bacterium Clostridium botulinum—the same toxin that causes a life-threatening type of food poisoning—being used to smooth their fine lines and wrinkles.
A small percentage of people are also toxin resistant, and there is a possibility of antibodies to the toxin developing over time, which may lead to decreased efficacy. In rare cases, a minute quantity of toxin may spread to adjacent areas, but this has been shown to be reversible. Botulinum toxin is also contraindicated in patients with a known allergy or hypersensitivity reaction to human albumin; women who are pregnant or breast-feeding; and in individuals with neurological diseases, such as multiple sclerosis, myasthenia gravis, and Eaton-Lambert syndrome.
The main effect of BTX-A is on muscle contraction because of its binding to the presynaptic nerve terminals, thus inhibiting the release of acetylcholine. Attempts to mimic the effects of injectable toxins are not new. We have seen countless topical formulations containing various peptides, most notably acetyl hexapeptide-3, marketed as Argireline, that offer short-term line-smoothing effects at best. What is new, however, are the recent advancements in the pike. From a topical neurotoxin to the use of focused cold energy and radiofrequency, it’s safe to say that injectable neurotoxins will no longer be the only game in town.
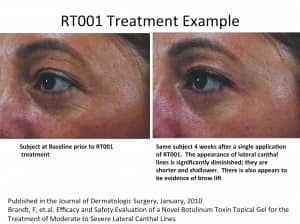
TOPICAL BOTOX?
Revance Therapeutics, Newark, Calif, which had a successful initial public offering in 2014, is gearing up for its first product, a topical form of BTX-A that may hold a broad list of potential indications.
Revance’s proprietary synthetic transport peptide is called TransMTS®. Its lead product, RT001, is a topical gel form of BTX-A in a single-use apparatus. It is applied to the skin and uses its proprietary peptide technology to deliver Botulinum toxin across the skin. It is currently in Phase 3 trials for crow’s feet.
“TransMTS is a family of peptides that drives the product into the skin by ‘tricking’ the cell into absorbing toxin complex and then transferring it to the other side,” explains Jacob Waugh, chief scientific officer and medical director of Revance Therapeutics. “The peptide is optimized to a mid-dermal delivery so that it captures what physicians are doing when they inject toxins. It can also be optimized for other uses, and we are exploring actives for OTC or other applications,” he says.
“Can a topical Botulinum toxin truly be effective? The answer is undoubtedly, yes,” says Brian Biesman, MD, a Nashville-based oculoplastic surgeon who was part of the Phase 2b study of RT001 in the US. “Their delivery system will transport large molecules into the skin, and the potential implications are very broad. Revance is interesting from the efficacy standpoint, as the outcome data from the Phase 2 trials was very similar to that achieved from injectable toxins.”
Also in Revance’s pipeline is RT002, an experimental injectable toxin designed for longer duration and more targeted delivery to specific treatment sites. Revance recently completed a Phase 1/2 clinical trial and is planning an active comparator trial outside the US for erasing glabellar lines.
Revance plans to finish its clinical studies in 2016 and to file its Biologics License Application for a commercial launch hopefully in 2017.
THE BIG CHILL
iovera° from Myoscience, Redwood City, Calif, delivers Focused Cold Therapy™ as an attractive option for people who want to treat their dynamic forehead wrinkles without a toxin. Currently, it is approved in Europe and Canada for temporary wrinkle reduction, temporary pain reduction, and the treatment of dermatologic conditions. iovera° has also been cleared in the US for pain management and general surgical use. It is delivered via a new 1- x 55-mm Round Smart Tip, which is a single-insertion blunt tip cannula designed for minimal discomfort, higher safety profile, and less risk of side effects, for a wider range of patients. Treatment takes about 15 minutes.
iovera° avoids some of the stigmata of the toxins, says London-based plastic surgeon Angelica Kavouni, MD, FRCS. “iovera° gives a great result for forehead lines, and the new tip is well tolerated after local anesthesia with minimal bruising,” she says. “Patient selection is crucial with this technology. Relaxation of the frontalis may cause a heavy brow and eyelids, and it does not treat crow’s feet. It is best suited for patients in their 30s, patients who dislike arched eyebrows, and for men with deep horizontal lines.”
Biesman adds that neither iovera° nor Revance are setting out to compete with the injectable toxins. Rather, their aim is to expand the market. In his view, there is room for them to peacefully coexist.
“Based on experience outside the US, it is an innovative and promising technology that will likely have a very real role in the aesthetic market,” he tells Plastic Surgery Practice. “I think there will be a group of patients who will be interested in these treatments. The device is well-designed and elegant.”
WRINKLE-BUSTING RF DEVICES
Another category of toxin alternatives uses radiofrequency (RF) to provide similar effects. While neurotoxins block the release of a neurotransmitter to inhibit muscle activity, RF systems create a small thermal injury to zap wrinkles into submission.
NEUBELLE™ RF from Serene Medical, San Ramon, Calif, is the only bipolar RF device cleared by the US Food and Drug Administration for temporary immobilization of facial motor nerves that control the corrugator/glabellar and frontalis muscle. The treatment has been shown to relax the muscle and smooth wrinkles for
12 months or longer, and muscle function returns gradually. The fine probe singles out a specific nerve for a precise and customized treatment. Unlike commercially available toxins, NEUBELLE can only be used on the forehead. Treatment takes about 20 minutes, and it is tolerable with local anesthetic injections. Results are immediate with minimal downtime.
A new contender in this category is ThermiRase™ from Thermi Aesthetics, Southlake, Tex. It is a minimally invasive procedure that disables the nerves that control the muscles responsible for glabellar frown lines.
Learn more about the products mentioned in this article by visiting these websites:
Patient selection is key with ThermiRase, says David J. Holcomb, MD, a facial plastic surgeon in Sarasota, Fla. “Neurotoxin nonresponders, and patients who object to neurotoxin treatment based on the recurring expense, recurring investment of time, concern about side effects, etc, are the ideal candidates for ThermiRase,” he says. “It is an excellent niche minimally invasive treatment with the potential to bring the aforementioned groups of patients into (or back into) your practice.’
In Holcomb’s experience, the optimum protocol is a single treatment. “The treatment takes up to an hour. The full effect may last more than a year, and there may be only incremental or partial return of function to corrugator/procerus muscles,” he says.
Wendy Lewis is president of Wendy Lewis & Co Ltd, Global Aesthetics Consultancy, www.wendylewisco.com, founder/editor in chief of beautyinthebag.com, and a contributing editor to Plastic Surgery Practice. She can be reached at [email protected].
Original citation for this article: Lewis W. It’s faux tox time. Plastic Surgery Practice. 2014;(8),36-37.