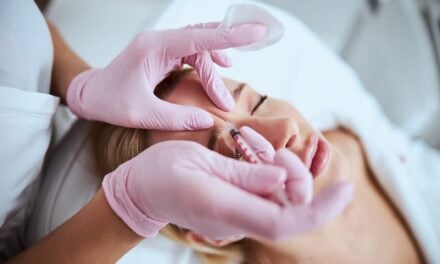
The U.S. FDA is issuing a warning about the potential risks associated with using fat-dissolving injections that are not FDA approved. These injections, also known as injection lipolysis, involve administering a series of injections under the skin to break down fat cells in specific areas. While FDA-approved options are available for reducing fat under the chin, the use of unapproved fat-dissolving injections poses serious health hazards, the FDA says.
Fat-dissolving injections not approved by the FDA are being sold under various brand names, including Aqualyx, Lipodissolve, Lipo Lab, Kabelline, and others. These products claim to reduce fat deposits in areas such as the chin, back, thighs, upper arms, and stomach. They often contain ingredients like phosphatidylcholine (PPC) and sodium deoxycholate (DC), sometimes referred to as “PCDC injections.” The FDA has not evaluated the safety or effectiveness of these unapproved ingredients, the agency says.
Reports have surfaced of consumers experiencing adverse reactions after using unapproved fat-dissolving injections, including permanent scars, serious infections, skin deformities, cysts, and painful knots. Some individuals received these injections from improperly licensed personnel, while others purchased the unapproved drugs online and self-administered them, leading to severe health complications.
Improper injection techniques, coupled with the use of unapproved injections, increase the risk of scarring, skin infections, and other serious complications, the FDA says. Safe and effective use of these products depends on correct injection practices, including the right number and location of injections, proper needle placement, and appropriate administration techniques. Consumers are strongly discouraged from purchasing unapproved fat-dissolving ingredients or attempting self-injection.
The only FDA-approved fat-dissolving injectable drug is Kybella, containing deoxycholic acid. It is approved solely for reducing fat under the chin in adults. The FDA has evaluated Kybella for quality, safety, and effectiveness specifically for this area; it is not approved for use in any other body areas, the FDA says.
Consumers are urged to consult a healthcare professional to receive FDA-approved fat-dissolving injections and to avoid purchasing such products online for self-administration. Adverse reactions from fat-dissolving injections or any other medications can be reported via the FDA’s MedWatch program.
Featured image: Multiple infected knots at the injection sites on the upper arm of a woman who received injections with Lipodissolve, a drug that is not FDA approved, and sought treatment at a dermatology clinic one week after the injections. (Kaur, H, C Reyes-Barron, WH Sipprell, A Cameron, T Louie, PR Tsai, and G Scott, 2022)