|
| Figure 1. BLU-U PDT Illuminator. PHOTOS COURTESY OF JEROLD J. OLSON, MD |
Photodynamic therapy involves the use of light to activate a photosensitizer that is localized in diseased tissues. This results in the formation of cytotoxic compounds, which cause the destruction of the affected tissues.
Use of light therapy to treat skin lesions is not new. In the 1960s Photofrin, a mixture of porphyrins, was developed as an intravenous photosensitizer. This drug was used to treat tumors that could be directly illuminated, such as those of the gastrointestinal, genitourinary, and pulmonary tracts. The main drawback was cutaneous photosensitivity.1
Topical photodynamic therapy has been developed in an effort to treat precancerous and cancerous skin lesions in a nonsurgical manner, without the risk of systemic side effects. In the United States 5-aminolevulinic acid (ALA)—from Dusa Pharmaceuticals of Wilmington, Del—and the methyl ester of 5-aminolevulinic acid (MAL)—from Metvix, Photocure AS of Norway and Galderma of Fort Worth, Tex—are approved for treatment of actinic keratoses (AK).
In Europe, New Zealand, and Australia MAL is approved for the treatment of AK, and basal cell carcinomas with the use of red light.
MECHANISM OF ACTION
As a topical photosensitizer, ALA is converted by the neoplastic tissue into protoporphyrin (PpIX), which is photosensitive. There is preferential production of these porphyrins in neoplastic tissue compared to non-neoplastic tissue.
Effectiveness of treatment depends on the penetration of the ALA into the skin, localization in diseased tissue, and effectiveness of the photoillumination.2 The conversion of ALA into PpIX can be demonstrated by examining the skin with a Wood’s light, as PpIX will demonstrate fluorescence.
The photodynamic process is initiated with the application of light at a wavelength matching an absorbance of the photosensitizer. After illumination with an appropriate light source, the photoactive porphyrins are excited to a higher energy state and then transfer energy to molecular oxygen, yielding highly reactive singlet oxygen. The singlet oxygen causes direct cell killing, vascular shutdown, and host-mediated damage through the release of inflammatory and immune mediators. In situ tissue damage occurs through both apoptosis and necrosis.3
Depending on the size and thickness of the lesion, the local damage evolves for approximately 1 to 2 weeks after treatment, with healing over a comparable period.
Multiple light sources for treatment sources have been used. The optimum choice would be a device that produces one of the excitation peaks of the photosensitizer, can treat a large surface area, and is affordable.
Absorption peaks for porphyrins are at 410, 505, 540, 580, and 630 nm. Blue light (Blue-U, Dusa) is preferable for treatment of AK. This device emits light at approximately 417 nm, corresponding to one of the excitation bands of PpIX (Figure 1).
Red light devices such as the Aktilite lamp (Galderma) have an output in the 570-nm to 670-nm range. This longer wavelength penetrates deeper and may be more effective for the treatment of lesions such as basal cell carcinoma.
Pulse dye lasers with a light output of 595 nm (such as the Candela Vbeam laser) lies just outside the absorption band of the PpIX, possibly requiring higher concentrations of the photosensitizer in order to be effective.4 The incoherent light sources allow treatment of a larger area in a shorter period of time. IPL devices have also been used. Typically, these are used as part of a photorejuvenation program targeting both AK and generalized photodamage.5,6
INDICATIONS AND PATIENT SELECTION
The current indications for photodynamic therapy of the skin (PDT) are in the treatment of AK, superficial basal cell carcinoma, and squamous cell carcinoma in situ.
PDT is also used for the treatment of acne and cosmetic improvement in photoaged skin. It is especially useful in the treatment of AK. These precancerous lesions have a potential for malignant transformation. Five percent to 20% of all AK will progress to squamous cell carcinoma. The transformation rate per year is 0.25% to 16%. Because AK can be multiple and arise in a widespread area, other forms of therapy such as cryotherapy or 5-fluorouracil cream can be problematic.
 |
| Figure 2. Levulan Kerastick |
PDT can be especially useful in transplant patients who develop many AK, which may more frequently progress to squamous cell carcinoma
Cryotherapy is the most commonly used destructive method of treatment for AK that typically is used only for a limited number of lesions. Residual hypopigmentation of the treatment sites are frequently noted. Topical chemotherapy with 5-fluorouracil cream or imiquimod is also effective.7 These treatments can cover large areas but require prolonged treatment times, and symptoms of erythema, crusting, and ulceration are common and sometimes limit patient compliance.
Patient acceptance of PDT is high, and the cosmetic outcomes have been judged as good. In our practice, we currently limit treatment to patients with multiple superficial AK who would benefit from treatment of larger surface areas. Careful examination prior to treatment is required to eliminate the possibility of underlying malignancies, such as squamous cell carcinoma or melanoma.
A low threshold for biopsy is had with all immunocompromised or organ transplant patients. The treatment is contraindicated in patients with cutaneous photosensitivity at wavelengths of 400 nm to 450 nm, porphyria, known allergies to porphyrins, or with known sensitivity to any of the components of the Levulan Kerastick. Additionally, patients should be screened for prescription medications that may cause photosensitivity.
PROCEDURE
Nursing personnel or physician’s assistants can perform skin preparation, application of the ALA, and treatment with the light source. The skin is prepared with a gentle cleansing with acetone. Hyperkeratotic lesions may be gently debrided with a curette.
The ALA solution is applied to the skin with the use of an applicator. It is important to follow the manufacturer’s recommendation for application of the Levulan Kerastick. It is especially important to ensure complete breakage of the ampules and sufficient mixing of the ingredients prior to application (Figure 2, above).
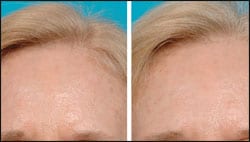 |
| Figure 3. A 48-year-old woman 6 weeks after receiving ALA-PDT therapy on her forehead for actinic keratosis. |
A variety of treatment protocols have been used. We favor application of the ALA approximately 1 hour prior to treatment. There is good evidence that an incubation period of 1 hour is sufficient for treatment of AK.8 If initial treatment is unsatisfactory, repeat treatments with longer incubation times and occlusive dressings to allow deeper penetration may be used.
We apply the ALA directly to the AK, followed by an application to the entire treatment area. This is based on the supposition there may be AK present too small to be visualized. After the incubation period, the patient is then placed in front of the light source. In our practice, we use blue light such as from the BLU-U from Dusa Pharmaceuticals, Wilmington, Mass.
The exposure lasts 16 minutes 40 seconds using the BLU-U at 10 mW/cm2. Both the patient and medical personnel must wear protective eyewear during the treatment. During the treatment, the patient will experience tingling and burning. We recommend the use of a cold air device such as the Syner-cool from Syneron Medical Ltd, Israel, to increase patient comfort. It is rare that a treatment will need to be interrupted secondary to patient pain.
As soon as the light exposure is completed, the discomfort will begin to subside. The patient is advised to avoid exposure to sunlight, sunlamps, or bright fluorescent lights for approximately 48 hours after therapy. They are informed that sunscreen will not provide protection from broad-spectrum light.
Patients are instructed to avoid spending any outdoor time for approximately 2 days after application. The scalp, extremities, and trunk can be easily protected with clothing during transportation home.
Typically, the treated lesions will develop erythema and edema. Most patients will have a moderate sunburn-type reaction that typically resolves within 7 to 10 days. Occasional crusting of the treated area can occur. Hyper or hypopigmentation has been reported, as well as scarring associated with treatment of skin cancers. Typically, we treat the face or scalp but do not treat these areas at the same time. This treatment can also be used in other areas of the body, especially the extremities. We have found a high degree of patient acceptance of this treatment; most patients with severe actinic damage have been willing to undergo multiple treatments.
RESULTS
Both ALA and MAL studies have shown good response in the clearance of actinic keratoses. Piacquido et al found that 77% of patients cleared 75% of their AKs at 8 weeks.9-11
Treatment with MAL and red light for basal cell carcinomas has revealed complete clearance rates of around 80%. Multiple authors note that recurrences of skin cancers can be seen up to 3 years.12-15 If there is a failure of PDT for treatment of skin cancers, it is reasonable to proceed to surgical excision with microscopic control of the margins.
SUMMARY
We feel that PDT with topical photosensitizers will have an increasing role in the treatment of skin disorders. Future avenues for research include the development of new photosensitizers that come with more affinity for cancerous and precancerous tissue.
An important role for PDT will likely be in skin cancer prevention.16 Additional uses may include an adjuvant to Moh’s micrographic surgery.17 We would like to see the establishment of accepted protocols for patients at high risk of non-melanoma skin cancers.
Katherine A. Orlick, MD, and Jerold J. Olson, MD, can be reached at Foothills Dermatology and Facial Plastic Surgery, Tucson, Ariz, at (520) 731-1110.
REFERENCES
- Zelickson BD. Mechanism of action of topical aminolevulinic acid. In: Goldman MP, ed. Photodynamic Therapy. Munich, Germany: Elsevier; 2005.
- Dougherty TJ, Gomer CJ, Henderson BW. Photodynamic therapy. J Natl Cancer Inst. 1998;90(12):889-905.
- Henderson BW, Dougherty TJ. How does photodynamic therapy work? Photochemo Photobiol. 1992;55:145-157.
- Braathen LR, Szeimies RM, Basset-Seguin N, et al. Guidelines on the use of photodynamic therapy for nonmelanoma skin cancer: an international consensus. International Society for Photodynamic Therapy in Dermatology, 2005. J Amer Acad Dermatol. 2007;56(1):125-143.
- Hall JA, Keller PJ, Keller GS. Dose response of combination photorejuvination using intense pulse light-activated photodynamic therapy and radiofrequency energy. Arch Fac Plast Surg. 2004;6:374-378.
- Marmur ES, Phelps R, Goldberg DJ. Ultrastructural changes seen after ALA-IPL photorejuvenation: A pilot study. J Cosmet Las Ther. 2005;7(1):21-24.
- Smith SR, Morhenn VB, Piacquadio DJ. Bilateral comparison of the efficacy and tolerability of 3% diclofenac sodium gel and 5% 5-fluorouracil cream in the treatment of actinic keratoses of the face and scalp. J Drugs Dermatol. 2006;5(2):156-159.
- Touma D, Yaar M, Whitehead S, Konnikov N, Gilchrest BA. A trial of short incubation, broad-area photodynamic therapy for facial actinic keratoses and diffuse photodamage. Arch Dermatol. 2004;140(1):33-40.
- Piacquadio DJ, Chen DM, Farber HF, et al. Photodynamic therapy with aminolevulinic acid topical solution and visible blue light in the treatment of multiple actinic keratoses of the face and scalp: investigator-blinded, phase 3, multicenter trials. Arch Dermatol. 2008;140(1):41-46.
- Rivers JK, Rosoph L, Provost N, Bissonnette R. Open-label study to assess the safety and efficacy of imiquimod 5% cream applied once daily three times per week in cycles for treatment of actinic keratoses on the head. J Cutan Med Surg. 2008;12(3):97-101.
- Fink-Puches R, Soyer HP, Hofer A, Kerl H, Wolf P. Long-term follow-up and histological changes of superficial nonmelanoma skin cancers treated with topical delta-aminolevulinic acid photodynamic therapy. Arch Dermatol. 1998;134:821-826;
- Wang I, Bendsoe N, Klinteberg CA, et al. Photodynamic therapy vs. cryosurgery of basal cell carcinomas: results of a phase III clinical trial. Br J Dermatol. 2001;144:832-840.
- Morton CA, MacKie RM, Whitehurst C, Moore JV, McColl JH. Photodynamic therapy for basal cell carcinoma: effect of tumor thickness and duration of photosensitizer application on response. Arch Dermatol. 1998;134:248-249.
- Wennberg AM, Lindholm LE, Alpsten M, Larko O. Treatment of superficial basal cell carcinomas using topically applied delta-aminolaevulinic acid and a filtered xenon lamp. Arch Dermatol Res. 1996;288:561-564.
- Soler AM, Werloe T, Berner A, Giercksky KE. A follow-up study of recurrence and cosmesis in completely responding superficial and nodular basal cell carcinomas treated with methyl 5-aminolaevulic-based photodynamic therapy alone and with prior curettage. Brit J Dermatol. 145:467-471.
- Wennberg A-M, Stenquist B, Stockfleth E, Keohane S, Lear JT, Jemec G. Photodynamic therapy with methyl aminolevulinate for prevention of new skin lesions in transplant recipients: A randomized study. Transplantation. 2008;86(3):423-429.
- Photodynamic therapy as adjuvant treatment of extensive basal cell carcinoma treated with Mohs micrographic surgery. Dermatol Surg. 2004;30(5):794-798.