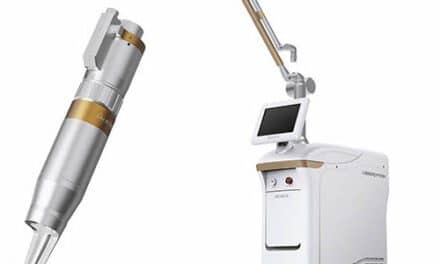
The Clear + Brilliant Touch laser, cleared by the FDA in 2022, includes two wavelengths designed to address wrinkles, pigmentation, and skin texture.
Bausch Health Companies Inc and its aesthetics business, Solta Medical, announced the launch of the Clear + Brilliant Touch laser in Canada. The device received approval from Health Canada on May 20, 2025.
The Clear + Brilliant Touch system builds on the Clear + Brilliant platform introduced in 2011. The device received US Food and Drug Administration clearance in 2022.
“The Solta business continues to expand its global presence through its diverse and well-established portfolio that supports the growing demand for high-quality aesthetics solutions,” says Thomas J. Appio, chief executive officer of Bausch Health, in a release. “Canada is an important part of that growth, reflecting strong and sustained interest in the aesthetic market across the country.”
The system uses fractional laser technology and allows clinicians to use two handpieces with different wavelengths during treatment. According to the company, the device is intended for use across a range of skin types and age groups and can be used year-round at the discretion of the treating physician.
“Expanding the availability of the Clear + Brilliant Touch System into Canada marks a significant step forward for Solta Medical and its commitment to global expansion of our innovative portfolio of products in skin health,” says Jiny Kim, senior vice president of Solta Medical at Bausch Health, in a release.
Two Handpieces for Dermatologic Indications
The system includes two fractional laser handpieces, each designed for specific dermatologic indications.
The 1440 nm handpiece is indicated for the treatment and improvement of wrinkles, dyschromia, pigmentation, skin texture, and pore size.
The 1927 nm handpiece is indicated for the treatment and improvement of wrinkles, dyschromia, pigmentation, and skin texture, as well as enhancement of skin permeability and pore size.
According to the company, the Clear + Brilliant platform has been supported by more than 15 clinical studies evaluating fractional laser technology for skin resurfacing and other dermatologic procedures.
Photo caption: Clear + Brilliant Touch laser
File photo / Bausch Health Companies Inc