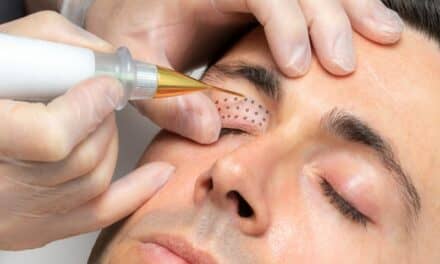
Dermalogica has launched the FDA-cleared PRO Pen Microneedling System, expanding into device-based treatments for acne scarring in adults.
Dermalogica has announced the launch of the PRO Pen Microneedling System, its first FDA-cleared device, marking the company’s entry into advanced in-office treatment technologies.
FDA Clearance and Indication
The PRO Pen Microneedling System is indicated for improving the appearance of facial acne scars in adults aged 22 years and older. According to the company, the clearance places the device among a limited number of microneedling systems to meet FDA regulatory standards for safety and performance in 2025.
Regulatory Expansion
In addition to U.S. clearance, Dermalogica has obtained regulatory authorizations in multiple international markets. The company reported achieving ISO 13485 certification in Canada prior to approval, and securing clearance in Australia through a review aligned with U.S. authorization. These steps were described as part of a broader effort to meet established regulatory frameworks across global markets.
Professional Availability and Positioning
Following FDA clearance, the device is now available to licensed skin health professionals through authorized channels. The company stated that the launch is intended to support integration between in-office procedures and at-home skincare regimens.
“As demand for minimally invasive, results-driven treatments like microneedling continues to grow, consumers are increasingly seeking personalized solutions that connect in-office care with at-home maintenance,” said Aurelian Lis, Dermalogica CEO. “Meeting those expectations is central to how we continue to innovate for providers and their patients.”
Device Design and Clinical Approach
The PRO Pen is designed for controlled skin penetration and is manufactured using biocompatible materials intended for safe contact with the skin. The company stated that the system is developed in accordance with medical device standards to support safety and procedural consistency.
“We’re encouraged by the progress we’ve made since receiving U.S. FDA 510(k) clearance for our 14-pin PRO Pen last fall, the response from the global professional skincare community and our dermatology partners has been extremely positive. When paired with our post-treatment Exo Booster system—featuring Lactobacillus-derived exosome-like vesicles—we’re seeing consistent, clinically relevant improvements in acne scarring, skin texture, and overall dermal quality across a wide range of skin types,” said Robert J. Bianchini, PhD, AAD.
Integration with Professional Portfolio
The device is positioned to integrate with Dermalogica’s existing professional treatments and homecare products. The company stated this approach is intended to support extended treatment plans, improve continuity of care, and create additional revenue opportunities through ongoing skincare regimens.
Industry and Practice Impact
Dermalogica described the launch as a milestone reflecting a broader shift toward clinically validated, minimally invasive treatments delivered within integrated care models.
The PRO Pen Microneedling System is now available through authorized professional channels.ices and in-clinic procedures.
Photo: Dermalogica