 |
It has been estimated that 10% to 20% of adults in the United States and Western Europe have varicose veins, and that up to 50% of women, by age 50, will have telangiectatic leg veins. The difference between varicose and telangiectatic leg veins is one of size. By convention, tortuous veins greater than 4 to 5 mm in diameter are referred to as varicose, veins between 1 and 4 mm in diameter are referred to as reticular, and veins less than 1 mm in diameter are referred to as telangiectasia.
The most common predisposing factor for the development of these unwanted leg veins is family history, but many other factors may also contribute to the development of varicose veins. Any activity or condition that increases abdominal pressure and impedes the flow of venous blood back to the heart may put strain on the veins, causing them to dilate.
These factors include standing for long periods of time, lack of exercise, obesity, constipation, wearing high-heeled shoes, and wearing tight undergarments or pants. The use of estrogen and/or progesterone hormone supplementation for birth control or postmenopausal symptoms also causes a dilatation of the venous wall. Localized trauma will initiate angiogenesis, with the eruption of telangiectasia.
Photodamage from the sun (or other forms of radiation) is also associated with an increased incidence of telangiectasia. Pregnancy puts strain on veins by increasing blood volume, increasing estrogen and progesterone levels, and impeding blood flow through compression of the pelvic venous system.
Varicose, reticular, and/or telangiectatic veins appear in one third of patients before age 25 and increase in incidence with age; 70% of people have visible cutaneous veins by age 70. For the most part, varicose and telangiectatic veins arise from (and may also be a cutaneous marker for) an underlying reversal of venous blood flow known as venous insufficiency. This reversal of blood flow produces venous hypertension in the lower extremities.
This, in turn, produces an increase in venous diameter that leads to valvular insufficiency. This results in a reversal of blood flow from the deep to the superficial venous system through incompetent communicating veins. The superficial veins respond to increased pressure by dilating to accommodate the increased blood flow.
In addition, with movement of the lower limbs, the high venous pressure that normally occurs within the calf-muscle pump is transmitted straight to the superficial veins and to the subcutaneous tissues drained by these communicating veins. When this occurs, the venous pressure in the cuticular venules increases and telangiectasia appears.
The point at which reflux occurs, whether through incompetent perforating veins, feeding reticular veins, or an incompetent saphenofemoral junction, must be treated first. Although endothelial cells are easily destroyed by chemical and physical insults (lasers and heat), they demonstrate a marked capacity for regeneration. The entire venous wall must be destroyed to destroy a vein completely.
A number of diagnostic tests can be performed to help the surgeon define the extent of varicose-vein disease and plan treatment. The primary goal is to locate the point of high-pressure reflux. This is easily accomplished using duplex ultrasound scanning, which allows visualization of the abnormal vein and its connections, as well as the determination of whether blood is flowing toward or away from the heart.1
Treatment
Depending on the cause of the problem, surgery, sclerotherapy, laser/intense pulsed light (IPL), or a combination of all techniques may be necessary. Surgical ligation and stripping procedures are essentially procedures of the past. For varicose veins more than 1 cm in diameter and for patients with an incompetent great saphenous vein or small saphenous vein, sclerotherapy is usually ineffective, except in the most expert of hands.
In such cases, very strong sclerosing solutions are necessary to destroy the vein, but a phlebitic reaction usually occurs, resulting in formation of a thrombus cord and overlying pigmentation of the skin. The procedure usually needs to be repeated two to four times, and even with initial closure of the vein, recurrence usually takes place within 5 years due to persistent high-pressure reflux.
Instead, I recommend endoluminal laser closure. This technique is performed under local tumescent anesthesia, with the patient being ambulatory immediately after the procedure. In my experience, it causes minimal (if any) pain, and patients can resume all normal activities within 24 hours. I recommend the 1,320-nm endoluminal laser, although other laser wavelengths and radiofrequency treatment are also used.2
For veins 4 to 10 mm in diameter, ambulatory phlebectomy is the treatment of choice. Veins of this size are removed through incisions of 2 to 3 mm, with no chance of recurrence (since the veins are on the surgical tray, not within the patient) and with a decreased risk of adverse sequelae.3
A beneficial effect of ambulatory phlebectomy is the harvesting of the veins, which are essentially collagen tubes. These can be transferred to areas that need filling, such as the nasolabial grooves and lips, saving the patient the cost of using temporary or artificial filling substances.4
Sclerotherapy is recommended for veins less than 4 mm in diameter. For veins less than 1 mm in diameter and/or for veins that persist after sclerotherapy and phlebectomy, laser or IPL treatment is recommended.1–5
Sclerotherapy Mechanisms
Sclerotherapy is the introduction of a foreign substance into the lumen of a vessel, causing thrombosis and subsequent fibrosis.1 Detergent sclerosants, such as sodium morrhuate, sodium tetradecyl sulfate, ethanolamine oleate, and polidocanol, produce endothelial damage through interference with the cell’s surface lipids.
Hypertonic-saline and hypertonic-glucose solutions produce dehydration of endothelial cells through osmosis, resulting in endothelial destruction. Chemical irritants or caustic agents such as glycerin and polyiodinated iodine produce direct destruction of the endothelial cells.
| Solution | Pigment | Allergy | Necrosis |
|---|---|---|---|
| Glycerin | – | – | – |
| Sodium tetradecyl sulfate | + | + | + |
| Polidocanol | + | + | + |
| Hypertonic saline | + | – | ++ |
The advantages and disadvantages of each solution are listed in Table 1. The recommended concentrations of sclerosing solution for various venous diameters are shown in Table 2.
Table 2. Recommended Solutions and Concentrations for Veins of Different Sizes
<1 mm:
- Glycerin
- Sodium tetradecyl sulfate, 0.25%
- Polidocanol, 0.5%
- Hypertonic saline, 11.7%
1 to 4 mm:
- Sodium tetradecyl sulfate foam, 0.25% to 0.5%
- Polidocanol foam, 0.5% to 1%
4 to 10 mm:
- Sodium tetradecyl sulfate foam, 1% to 3%
- Polidocanol foam, 2% to 5%
Sodium tetradecyl sulfate is the only sclerosing agent approved by the FDA, although hypertonic saline and glycerin solutions are also widely used for veins less than 1 mm in diameter. I do not advise using compounded detergent sclerosing solutions, since they have been found to contain a variety of contaminants that could cause both short-term and long-term health problems.6
Injection Technique
Sclerotherapy should progress from the largest to smallest vessels. The quantity of solution to be injected should be enough to fill the vessel and displace intravascular blood. When the solution stops flowing, the injection should end, as this means that the solution is flowing into the deeper venous system.
To avoid the risk of new telangiectasias forming around the edge of the treated area, the minimal effective sclerosing concentration should be used. This will minimize the resulting inflammation. It is important to remember that sclerotherapy is a controlled thrombophlebitic reaction. The entire venous system of each leg is treated at one time to avoid leaving areas of refluxing blood flow that will cause recanalization of the treated vessel or extravasation of red blood cells from the damaged vessel.
I recommend the use of a foamed sclerosing solution for all veins greater than 1 mm in diameter. Foaming can only be done with a detergent sclerosing solution such as sodium tetradecyl sulfate. I use 1 mL of solution to 4 mL of air. Foaming a sclerosing solution doubles its effective sclerosing power while decreasing its adverse-effect profile by a factor of four.1,7,8
Before & After |
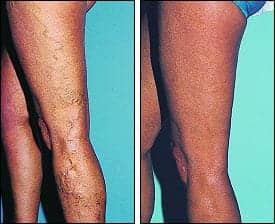 |
| Figure 1. A 56-year-old woman with a varicose vein on her lateral thigh is shown before and 2 years after treatment with 11 mL of 1% sodium tetradecyl sulfate compression sclerotherapy. |
The patient is told to walk immediately after the session to help prevent deep-vein thrombosis and to constrict the superficial and perforating veins. Calf-muscle movement produces a rapid blood flow in the deep venous system that dilutes any sclerosant that might have migrated to this area. Following injection of all varicose or telangiectatic veins, they are compressed to minimize thrombosis.
Compression
Postsclerosis compression, described in the 1950s and 1960s, may be the most important advance in sclerotherapy for varicose veins since the introduction of relatively safe synthetic sclerosing agents in the 1940s. Primarily, compression eliminates a thrombophlebitic reaction and substitutes sclerophlebitis, with the production of a firm, fibrous cord. Compression serves at least five purposes. First, compression, if adequate, may result in direct apposition of the treated venous walls to produce more effective fibrosis. Therefore, sclerosing solutions of lower strength may be used successfully. Second, compressing the treated vessel will decrease the extent of thrombus formation that inevitably occurs with the use of all sclerosing agents, thus decreasing the risk of recanalization of the treated vessel.
Third, a decrease in the extent of thrombus formation may also decrease the incidence of postsclerosis pigmentation. Fourth, the limitation of thrombosis and phlebitic reactions may prevent the appearance of telangiectatic matting. Fifth, the function of the calf-muscle pump is improved by the physiologic effect of a graduated compression stocking.
In addition, by externally supporting untreated large veins, compression stockings will narrow their diameter, restoring competency to valvular function (which decreases retrograde blood flow). External pressure will also retard the reflux of blood from incompetent perforating veins into the superficial veins. In short, compression sclerotherapy is now the standard practice for the treatment of varicose veins.
Microsclerotherapy
In the vast majority of cases, spider veins are in direct connection with underlying varicose veins, either directly or through tributaries. Therefore, as for varicose veins, treatment should first be directed to plugging the leaking high-pressure outflow at its point of origin.
Think of spider veins as the fingers and of the feeding varicose vein as the arm. Treatment should first be directed to the feeding arm and, only if necessary, directly to the spider fingers.
There are a number of advantages to this systematic approach to sclerotherapy. The spider veins often disappear without direct treatment, thus limiting the number of injections needed. Also, the larger feeding vein is both easier to cannulate and less likely to rupture upon injection of the sclerosant, thus minimizing the extent of extravasated red blood cells and sclerosant. Even the compression of leg veins less than 1 mm in diameter has been shown9 to minimize the development of postsclerotherapy hyperpigmentation, cutaneous necrosis, and telangiectatic matting.
Table 3. Complications and Adverse Sequelae of Sclerotherapy
- Hyperpigmentation
- Telangiectatic matting
- Localized urticaria
- Tape-compression folliculitis
- Vasovagal reflex
- Cutaneous necrosis
- Arterial injection
- Superficial thrombophlebitis
- Pulmonary embolism and deep-vein thrombosis
- Temporary swelling
- Pain with injection
- Tape-compression blister
- Recurrence
- Localized hirsutism
- Allergic reaction
- Nerve damage
Microsclerotherapy for spider veins is performed using a standard technique. The patient is placed in a supine position. Gravitational dilatation of telangiectasias is unnecessary. A loupe for magnification is helpful to aid visualization. The goal of microsclerotherapy is to cannulate the vessel so that the sclerosant will be deposited within, and not outside, the vessel wall. A 30-gauge needle is sufficient.
For vessels less than 1 mm in diameter, a 72% glycerin solution, mixed at a two-to-one ratio with 1% lidocaine with epinephrine, is best. With this sclerosing solution, there is virtually no risk of ulceration, pigmentation, or telangiectatic matting. In addition, resolution of the veins appears to be better than that seen with use of a detergent solution.10
Course and Prognosis
Patients should be examined 2 weeks after injection so that any area of thrombosis can be evacuated early. The treated area should not be retreated sooner than 6 to 8 weeks after injection, to allow for resolution of inflammation between treatments.
Unfortunately, sclerotherapy, like any therapeutic technique, has with it a number of potential adverse sequelae and complications (Table 3). Fortunately, these complications are quite rare. Fairly common adverse sequelae include temporary perivascular cutaneous pigmentation and a temporary flare of new perivascular telangiectasias. Relatively rare complications include localized cutaneous necrosis, thrombophlebitis of the injected vessel, arterial injection with resultant distal necrosis, and pulmonary emboli. These complications and ways to minimize them are discussed in detail elsewhere.1
Mitchel P. Goldman, MD, is a volunteer clinical professor of dermatology/medicine, University of California, San Diego. He can be reached via his Web site, www.Spa-MD.com.
References
- Goldman MP, Bergan JB, Guex JJ. Sclerotherapy Treatment of Varicose and Telangiectatic Leg Veins: Diagnosis and Treatment. 4th ed. London: Mosby-Elsevier; 2007.
- Goldman MP, Mauricio M, Rao J. Intravascular 1320nm laser closure of the great saphenous vein: a 6–12 month follow-up study. Dermatol Surg. 2004;30:1380–1385.
- Ricci S, Georgiev M, Goldman MP. Ambulatory Phlebectomy: A Practical Guide for Treating Varicose Veins. 2nd ed. Philadelphia: Marcel Dekker; 2005.
- Blugerman G, Goldman MP. Autologous vein/collagen transplantation for correction of dermal atrophic changes. Dermatol Surg. 2002;28:372–375.
- Goldman MP. Cosmetic and Cutaneous Laser Surgery. London: Mosby-Elsevier; 2006.
- Goldman MP. Sodium tetradecyl sulfate for sclerotherapy treatment of veins: is compounding pharmacy solution safe? Dermatol Surg. 2004;30:1454–1456.
- Barrett JM, Allen B, Ockelford A, Goldman MP. Microfoam ultrasound guided sclerotherapy of varicose veins in 100 legs. Dermatol Surg. 2004;30:6–12.
- Rao J, Wildemore JK, Goldman MP. Double-blind prospective comparative trial between foamed and liquid Aethoxysklerol and Fibrovein in the treatment of varicose and telangiectatic leg veins. Dermatol Surg. 2005;31:631–635 .
- Weiss RA, Sadick NS, Goldman MP, Weiss MA. Post-sclerotherapy compression and its effects on clinical outcome. Dermatol Surg. 1999;25:105–108.
- Leach B, Goldman MP. Comparative trial between sodium tetradecyl sulfate and glycerin in the treatment of telangiectatic leg veins. Dermatol Surg. 2003;29:612–625.


