8/18/08
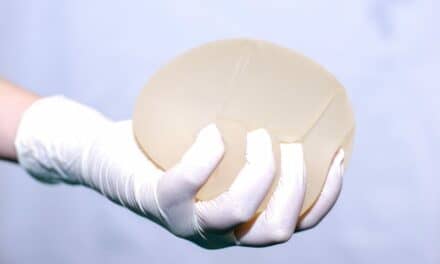
Evera Medical announces that the U.S. Food and Drug Administration has granted conditional approval of the company’s investigational device exemption (IDE) application for its FulFil Lip aesthetic augmentation implant. The approval allows Evera Medical to assess the safety and effectiveness of the FulFil Lip implant in patients undergoing aesthetic lip enhancement. Enrollment into the study, named EIFFEL (Evera Implant—FulFil—For Enhancement of the Lip), is expected to begin within 30 days.
FulFil Lip consists of a very thin, stretchy balloon, which when filled with saline, is designed to replicate the supple, natural look and feel of youthful lips. The implant is adjusted until the desired degree of enhancement is achieved. Once the implant is placed in the lip, a micro-valve prevents fluid from escaping. A thin coating allows gentle attachment to the lip tissue, without excessive scarring or firmness.
“In formulating EIFFEL, we worked closely with the FDA, who reviewed our extensive bench and biocompatibility testing along with the long-term outcomes of our international lip augmentation study,” says Dr. Michael D. Lesh, CEO of Evera Medical. “We are quite pleased with this approval and are optimistic that the trial endpoints will be met. FulFil Lip will be the first treatment of any kind to be specifically labeled for lip enhancement.”
[Medical News Today, August 13, 2008]