 |
Over the past 3 decades, microsurgery has proven itself a reliable technique for the reconstruction of complex defects in all locations of the body. Once limited to university centers, microsurgery is now performed in community hospitals by those specially trained in microsurgical techniques.
The surgeon’s ability to consistently transfer skin, muscle, fascia, and bone has allowed single-stage reconstruction of composite defects of the head and neck, breast, trunk, and extremities. This has translated to faster recovery times, cost savings, and aesthetically pleasing results.
Refinements in techniques, such as perforator flaps, have allowed surgeons to achieve cosmetically successful results while minimizing donor-site morbidity.
One of the most demanding and challenging sites of reconstruction is the head and neck. Due to the aesthetic and functional requirements of the oral cavity, oropharynx, and hypopharynx, close collaboration with our colleagues in head-and-neck surgery, oral surgery, dentistry, and speech therapy is required for a successful outcome.
Nonmicrosurgical alternatives for soft tissue include the pectoralis major flap, the deltopectoral flap, and the platysma flap. Nonmicrosurgical options for bone reconstruction include bridging reconstruction plates, bone trays, and nonvascularized bone grafts. This type of reconstructive approach has certain limitations, especially in an irradiated field, or in a patient who will require adjuvant radiation. The limitations include plate fracture, plate exposure, excessive bulk, and decreased options for dental restoration.
The advantage of transferring a virtually unlimited amount of pliable, well-vascularized sensate and composite tissue is specific to microvascular reconstruction. Additionally, the ability to reconstruct “like tissue with like tissue” is another appealing advantage.
FIRST THINGS FIRST
Prior to establishing a microsurgery program, a commitment on the part of the surgeon and hospital is required to ensure success. It is essential to in-service the operating room, anesthesia, and nursing staffs before proceeding. I employ a physician’s assistant who assists both intraoperatively and with postoperative management. She acts as a liaison between the nursing staff, rotating residents, and consulting services. Furthermore, she has proven to be a cost-effective addition to the surgical team.
 |
Before embarking upon a complex reconstruction, detailed preoperative planning is required. Appropriate planning helps to avoid intraoperative surprises and assists with coordination of the surgical team. A preoperative medical evaluation is required, with particular attention to the patient’s cardiopulmonary status.
Additional imaging studies are dictated by the patient’s medical condition and the type of flap performed. In the elderly population, a lower-extremity arteriogram or CT-angiogram is typically performed when a fibular donor site is chosen. This assesses the degree of atherosclerosis as well as the potential for a peroneal dominant circulation.
The next step requires an understanding of the primary tumor and the extent of the planned resection. These details include knowledge of not only tumor stage and prognosis, but also an understanding of the patient’s functional status and goals.
The goals of head-and-neck reconstruction include protection of vital structures and stable wound closure, as well as the restoration of both form and function. Functional requirements of the oral cavity, oropharynx, and hypopharynx must be appreciated and include oral competence, articulation, and deglutition.
 |
 |
| Figure 1. Preoperative view of a 50-year-old male with osteoradionecrosis of the mandible refractory to hyperbaric oxygen and antibiotics. Sevenmonth postoperative view after fibula osseocutaneous flap. | |
A checklist is created in order to take an inventory of the reconstructive requirements. This checklist is then compared to the advantages and disadvantages of the particular flaps.
Assessment begins by dividing defects into the following three categories:
- Those resulting from primary surgical extirpation;
- Those that result from surgery for local recurrence after failed radiation therapy; and,
- Defects due to complications of radiation therapy.
As the role of chemoradiation for the primary treatment of these malignancies has expanded, reconstruction in irradiated fields has become more commonplace. In these defects, successful wound closure and primary soft-tissue healing assumes increased importance. In irradiated fields, successful soft-tissue closure may take precedence over restoring bony continuity.
In addition, osteoradionecrosis of the mandible is an infrequent but debilitating complication of radiation therapy. Treatment requires resection of the affected bone and either bone or soft-tissue reconstruction.
 |
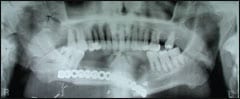 |
| Figure 2. Preoperative panorex demonstrating bony resorption and postoperative panorex obtained prior to discharge from the hospital. |
Figure 1 demonstrates osteoradionecrosis of the mandible and floor of the mouth treated with composite resection and reconstruction with a fibula osseocutaneous flap.
When possible, two preoperative office visits are scheduled with both the patient and family. The procedure is explained in detail, and photographs of donor sites—especially the radial forearm, anterolateral thigh, and fibula—are shown. The limitations of functional results are discussed. This includes the possibility of postoperative aspiration and the need for subsequent laryngeal suspension or laryngectomy, the need for speech therapy, and the possibility of prolonged tube feeds if the patient is unable to maintain adequate caloric intake. The potential for flap failure and the possibility of reoperation are also reviewed. Finally, nonmicrosurgical options are outlined.
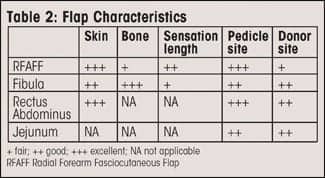
After the nature of the defect has been determined, the appropriate flap must be selected. Typically, variations on several flaps are chosen. The choice of flap largely depends upon two factors: the need for soft tissue and bone, as well as the ratio and location of soft-tissue requirements to bony requirements (Table 1). These include the radial forearm flap, the anterolateral thigh flap, the rectus abdominis flap, the jejunal flap, the fibula flap and, less frequently, the iliac crest and scapular flaps.
Soft-tissue-only defects of the oral cavity are most commonly reconstructed with either a radial forearm fasciocutaneous flap or, if larger volumes are required, with the anterolateral thigh or the rectus abdominis flap. In cases of a hemiglossectomy, the forearm is able to provide a sensate reconstruction by approximating the lateral or medial antebrachial cutaneous nerve of the forearm with the lingual nerve. Figure 3 demonstrates reconstruction of a hemiglossectomy defect with a radial forearm flap. In addition, if a short, lateral mandibular segmental defect is involved, a segment of radius may be incorporated in the forearm flap.
 |
 |
 |
Figure 3. A: Intraoperative view of a 69-yearold male with squamous cell carcinoma of the tongue undergoing hemiglossectomy and reconstruction with a radial forearm fasciocutaneous flap. B: Five-month postoperative view of forearm donor site. C: One-year postoperative view of left tongue reconstruction with forearm flap. |
A potential pitfall of radial bone harvest is the risk of fracture. As the requirements for bony reconstruction are increased, the fibular flap is often utilized. The defect is characterized as lateral mandible, anterior mandible, hemi-mandible, and total mandible. Reconstruction with vascularized bone is of greater clinical importance if the defect is larger than 5 cm or located in the anterior mandible, due to the increased loading forces. The fibula allows total mandibular reconstruction with multiple osteotomies, which remain vascularized by the periosteal circulation.
Other notable, but less frequently used, composite free flaps include the iliac crest and the osteocutaneous scapular flaps. Although the quantity (and quality) of the scapula may be less when compared to the fibula, it may provide greater skin coverage. Whereas the iliac crest provides a large amount of bone naturally shaped in the form of a hemi-mandible, the potentially unreliable and immobile skin paddle, as well as the risk of donor-site hernia formation, has limited the use of this flap in our practice. Osseointegrated dental implants may be placed as part of the primary procedure or as a secondary procedure.
For large-volume, soft-tissue-only reconstruction, the rectus abdominis flap or the anterolateral thigh flap may be employed. These flaps may be utilized in total glossectomy defects or large, previously irradiated fields. Finally, for reconstruction of the pharynx, options include the radial forearm, anterolateral thigh, or jejunal flaps. These defects may be categorized as partial versus complete pharyngeal defects.
 |
For complete, circumferential defects, a tubed radial forearm or anterolateral thigh or free jejunal flap may be utilized. As these are buried flaps, a monitoring segment is designed and exteriorized in the neck. When using a jejunal flap, a separate jejunal segment based on the same vascular arcade can be externalized and monitored. Furthermore, internal implantable Doppler devices can be used for flap monitoring as well. Figure 4 demonstrates a patient undergoing laryngopharyngectomy and bilateral neck dissections reconstructed with a tubed radial forearm flap. (Detailed discussions regarding voice rehabilitation and swallowing are beyond the scope of this article.)
A second important determinant in flap choice is the availability of recipient vessels. The usual choice of vessels is branches of the ipsilateral external carotid artery, or an end-to-side anastomosis to the external carotid artery itself, and the ipsilateral internal or external jugular vein.
Figure 5 demonstrates resection of the soft palate and lateral pharyngeal wall with a radial forearm reconstruction. Ideally, the anastomosis is performed to nonirradiated vessels. Depending upon the patient’s radiation history, this may require recipient vessels in the contralateral neck.
 |
 |
| Figure 4. Intraoperative view of a 51-year-old female with postradiation, recurrent squamous cell carcinoma of the left oropharynx. | Figure 5. Intraoperative view of a specimen from a 61-year-old male with squamous cell tumor of the tongue and the floor of the mouth. |
 |
 |
| Figure 6. Intraoperative view of a specimen from a 56-year-old male with high-grade adenocarcinoma of the right submaxillary gland involving the mandible, cheek, and marginal mandibular nerve. | Figure 7. Intraoperative view of right fibula osseocutaneous flap in situ with reconstruction plate in place. |
A distinct advantage of the radial forearm flap is the long, reliable vascular pedicle. If recipient vessels in the neck are unavailable, the ipsilateral thoracoacromial pedicle may be used. These recipient vessels may be utilized even if a prior pectoralis major flap has been performed (Table 2, page 26).
THE PROCEDURE
In most circumstances, a two-team approach is utilized in order to decrease operating time. When positioning the patient, it is helpful to rotate the bed 180°. This places the anesthesiologist at the patient’s feet and allows space for the microscope and two surgical teams.
The head-and-neck surgeon begins by performing the resection. The access incision depends upon the location of the lesion. Three standard incisions are used. These include an intraoral approach, an apron incision, or a lip-split with mandibulotomy.
Although the lip-split approach allows ease of insetting in the posterior oropharynx, an apron incision is preferable when possible. In general, this results in a less conspicuous scar. Figure 6 demonstrates a patient undergoing glossectomy and floor-of-mouth resection through an apron incision.
As soon as a template of the defect is developed, flap elevation begins. In certain circumstances, flap elevation commences simultaneous with the resection. In these instances, final flap design can be determined once the resection is completed. Flap elevation is performed with the help of a physician’s assistant or resident, and the microvascular anastomoses are performed by the reconstructive surgeon with the assistance of the head-and-neck surgeon, resident, or physician’s assistant. One time-saving approach for the venous anastomosis is the use of the microvascular coupler. Operative efficiency is improved by having the physician’s assistant close the donor site while the anastomosis and insetting are being performed.
Despite the increased prevalence of microsurgery, there remains little consensus regarding the use of anticoagulation. Protocols range from aspirin only to systemic anticoagulation, such as heparin and dextran infusions. At present, in our practice, a 300-mg aspirin suppository is administered on induction and 5,000 units of heparin are given subcutaneously for prophylaxis against deep-vein thrombosis. Systemic heparinization is occasionally utilized prior to the anastamosis. This is generally reserved for cases in which anastamoses will be performed to radiate vessels.
A heparin solution at a concentration of 500 U/cc is used for intraluminal irrigation. Postoperatively, 81 mg of aspirin are administered daily, and enoxaprin is given twice daily for DVT prophylaxis. The aspirin is continued for 2 weeks postoperatively.
Over the past 7 years, a dextran infusion has been administered on one occasion. This case involved acute and recurrent intraoperative arterial thrombosis. A test dose is administered to assess for an allergic reaction, and the infusion is run at 40 cc per hour. No consensus exists on the length of therapy.
A variety of modalities are utilized for postoperative monitoring. These include external Dopplers, implantable Doppler probes, and external tissue oxygen-saturation monitors. However, the most reliable method of flap monitoring may be clinical evaluation by a trained observer.
As most flap failures occur in the first 24 to 48 hours after surgery, frequent assessment by the nursing staff is performed. This entails assessment at 15-minute intervals for the first 6 hours postoperative, 30-minute intervals for the next 6 hours, followed by hourly assessment for the next 72 hours. The majority of these patients are monitored in the intensive care unit during this period. Flap checks document the presence of an arterial signal by external Doppler, color, temperature, and turgor. I am contacted directly for any concern of flap viability or venous congestion. In addition, the tracheostomy tube is sutured to the neck skin. Tracheostomy ties and compression dressings are avoided in the early postoperative period in order to minimize the risk of pedicle compression.
When a skin graft is required for the forearm, fibula, or anterolateral thigh donor site, a negative-pressure dressing and posterior splint or knee immobilizer are placed. The negative-pressure dressing serves as an excellent bolster for the skin graft. This dressing is removed on postoperative day 4. Subsequent dressings involve every other day dressing changes with nonadherent gauze and a light, compressive wrap. Ambulation begins between postoperative day 5 and 7 when a skin graft is placed on a lower-extremity donor site.
Many patients will benefit from some form of enteric feeding tube prior to surgery. Often, a percutaneous endoscopic gastrostomy is placed preoperatively by the gastroenterology service. For less-involved procedures, a nasogastric tube placed at the time of surgery may suffice in the early postoperative period, until the patient is able to resume an oral diet. If the patient is unable to maintain adequate oral intake, a PEG tube or open gastrostomy tube may be placed postoperatively.
Furthermore, a tracheostomy is commonly employed for more complex intraoral procedures. If adjuvant radiation therapy is required, the tracheostomy is downsized and converted to a cuffless tube prior to discharge. Otherwise, the patient is decannulated prior to discharge from the hospital.
Oral intake is initiated 7 to 10 days postoperatively, after evaluation by speech therapy. For reconstruction of the pharyngoesophagus or base of tongue, a swallow study is performed on postoperative day 7 to 10 in order to assess swallowing physiology and check for an enteric leak.
|
See also “Vectors of Facial Aging and Their Reversal” by Joseph Niamtu III, DMD, in the June 2008 issue of PSP. |
 |
Many patients are able to maintain adequate caloric intake with oral feeding. If they are unable to do so, they are discharged with tube feeds. Depending upon the nature of the procedure, the patient is discharged on postoperative day 10 to 14. If adjuvant radiation therapy is required, this is typically initiated 6 weeks after surgery. For one patient with adenocarcinoma of the submaxillary gland invading the marginal mandibular nerve and cheek, a composite resection was performed. The defect was reconstructed with a fibula osseocutaneous flap. Adjuvant radiation therapy was initiated 6 weeks postoperatively.
CONCLUSION
Microsurgical reconstruction of complex head-and-neck defects allows effective, single-stage reconstruction of even large, composite defects. These techniques are cost-effective modalities and offer distinct advantages over traditional, nonmicrosurgical techniques. With a committed surgeon and staff, highly successful results can be expected.
Loren Schechter, MD, is a plastic surgeon based in Morton Grove, Ill. He serves as the chief of plastic surgery at Chicago Medical School and Division Director of Plastic Surgery at Lutheran General Hospital in Park Ridge, Ill. He can be reached via e-mail at .
Iris Seitz, MD, PhD, is a senior resident in plastic surgery at the University of Chicago.



