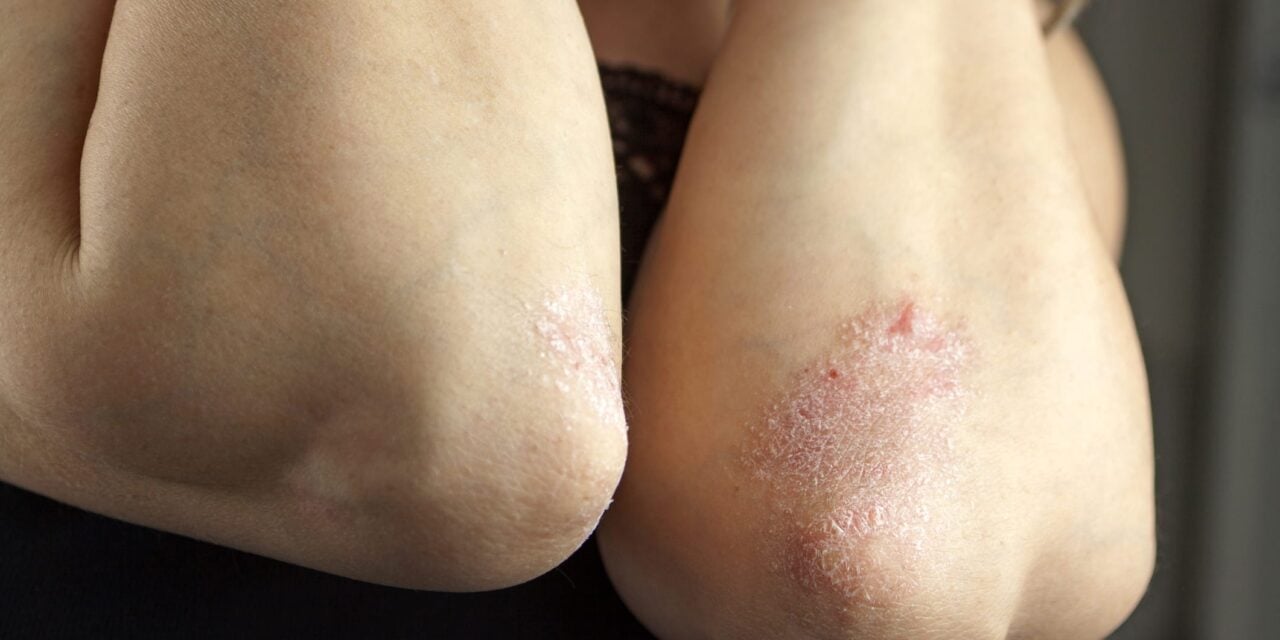
Approval is based on Phase 3 data showing high rates of skin clearance in patients eligible for systemic treatment or phototherapy.
Johnson & Johnson announced that the US Food and Drug Administration (FDA) has approved Icotyde (icotrokinra), an interleukin-23 (IL-23) receptor antagonist, for the treatment of moderate-to-severe plaque psoriasis in adults and pediatric patients aged 12 years and older who weigh at least 40 kg and are candidates for systemic therapy or phototherapy.
According to the company, Icotyde is an oral peptide designed to block the IL-23 receptor and is administered as a once-daily pill.
“Icotyde delivers something unique in psoriasis treatment, combining skin clearance with a favorable safety profile in a once–daily pill, making it an easy addition to a patient’s routine,” says Linda Stein Gold, MD, director of dermatology clinical research at Henry Ford Health, in a release. “With new guidance from the International Psoriasis Council that clarifies when to move beyond cycling on topical treatments to systemic therapy, an innovative option like Icotyde is a potential game–changer for many adult and adolescent patients.”
Clinical Data
The FDA approval is supported by results from four Phase 3 studies involving approximately 2,500 patients. The ICONIC clinical development program evaluated Icotyde in adults and adolescents, including patients with psoriasis affecting high-impact areas such as the scalp and genital regions, as well as in head-to-head trials against an active comparator.
In those studies, approximately 70% of patients achieved clear or almost clear skin (Investigator’s Global Assessment 0/1), and 55% achieved a Psoriasis Area and Severity Index 90 response at Week 16. Rates of adverse reactions were within 1.1% of placebo through Week 16, and no new safety signals were identified through Week 52.
“With the FDA approval of Icotyde, Johnson & Johnson is setting a new standard for the treatment of moderate-to-severe plaque psoriasis,” says Jennifer Taubert, executive vice president, worldwide chairman, innovative medicine, Johnson & Johnson, in a release. “We’re proud to bring this game-changing innovation to the market, marking a transformative shift in plaque psoriasis management that empowers patients and clinicians to reach their treatment goals.”
Disease Burden and Treatment Considerations
Psoriasis affects more than 8 million people in the United States and can impact physical comfort and quality of life, particularly when lesions appear on visible or sensitive areas. For patients with moderate-to-severe disease, systemic therapies are often used when topical treatments do not provide sufficient improvement.
Guidance from the International Psoriasis Council recommends considering systemic therapy if two cycles of topical medications, each applied for four weeks, do not lead to meaningful improvement.
“Finding the right treatment can take time, during which people with psoriatic disease should be considering multiple factors from efficacy to safety to how the treatment fits into their everyday life,” says Leah M. Howard, JD, president and CEO of the National Psoriasis Foundation, in a release. “The approval of a novel systemic therapy changes the conversation about treatment options for our community.”
John Reed, MD, PhD, executive vice president, R&D, innovative medicine, Johnson & Johnson, adds in a release, “The approval of Icotyde represents a pivotal moment for people with plaque psoriasis. At Johnson & Johnson, we are harnessing our scientific expertise to transform cutting-edge science into meaningful solutions for patients. Icotyde is a fundamentally different treatment with the potential to redefine what physicians and patients can expect from psoriasis treatment.”
Access and support
Johnson & Johnson said it will offer a patient support program, Icotyde withMe, which includes resources such as cost support options, access to a nurse guide, and educational materials for patients prescribed the therapy.
ID 41100431 © Hriana | Dreamstime.com