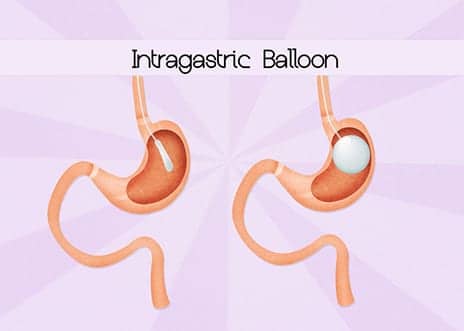
The FDA recently issued a Letter to Health Care Providers detailing risks for hyperinflation and acute pancreatitis observed in post-approval studies for two liquid-filled intragastric balloons indicated for weight loss, both approved in 2015 and manufactured by Apollo Endosurgery.
In the studies, 2.3% (6 of 258) of participants using the Orbera balloon system experienced hyperinflation of the balloon with events occurring from within a week of a placement to 23 weeks; 1.3% (2 of 159) of participants using the ReShape balloon system experienced acute pancreatitis — one participant at 3 days and the other at 2 weeks after placement. Participants who experienced the adverse events had symptoms of nausea, vomiting and abdominal pain; the devices were removed early for these participants. No instances of hyperinflation were observed with the ReShape system, and no cases of acute pancreatitis were observed with the Orbera system, according to the letter.