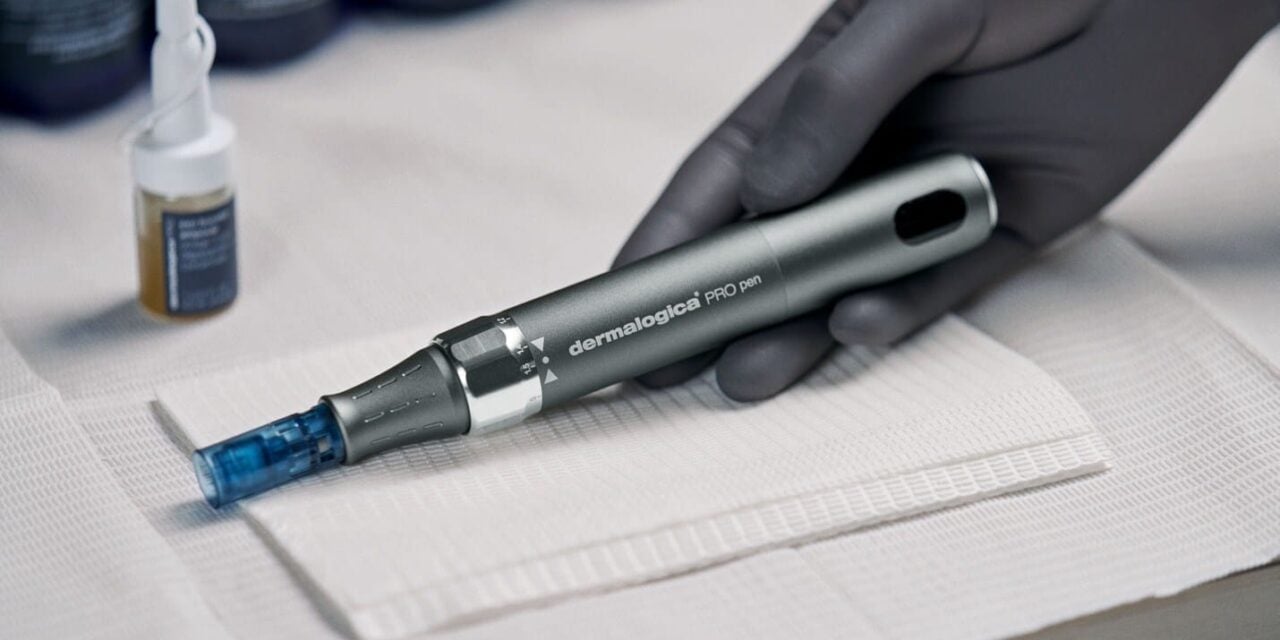
The PRO Pen is indicated for improving the appearance of facial acne scars in adults aged 22 years and older.
Dermalogica has announced the launch of its PRO Pen Microneedling System, the company’s first device to receive clearance from the US Food and Drug Administration (FDA). The system is intended for use as a treatment to improve the appearance of facial acne scars in adults.
The company also secured regulatory authorizations for the device in Canada and Australia. In Canada, ISO 13485 certification was obtained before device approval, establishing Dermalogica as a medical device manufacturer under international quality systems standards. According to a company release, these approvals demonstrate an investment in meeting regulatory frameworks in markets that require clinical oversight and specific device standards.
“Dermalogica has always been part of a professional ecosystem that bridges professional in-office treatments with at-home skincare,” says Aurelian Lis, Dermalogica CEO, in a release. “The PRO Pen extends this integration, enabling providers to design more comprehensive treatment journeys that support consistent outcomes while strengthening client loyalty and lifetime value.”
The PRO Pen is now available to licensed skin health professionals.
Device Features and Clinical Integration
The PRO Pen Microneedling System is manufactured using biocompatible materials and is validated for controlled skin penetration, according to the company. It is designed to integrate with the brand’s portfolio of professional treatments and at-home skincare products, allowing providers to connect in-office procedures with prescribed homecare regimens.
“We’re encouraged by the progress we’ve made since receiving US FDA 510(k) clearance for our 14-pin PRO Pen last fall. The response from the global professional skincare community and our dermatology partners has been extremely positive,” says Robert J Bianchini, PhD, AAD, in a release. “When paired with our post-treatment Exo Booster system—featuring Lactobacillus-derived exosome-like vesicles—we’re seeing consistent, clinically relevant improvements in acne scarring, skin texture, and overall dermal quality across a wide range of skin types.”
The company states this integrated approach can potentially amplify results while minimizing downtime.
“Dermalogica’s 40-plus year legacy in skin health is a key reason the Pro Pen was such a natural addition to my practice,” says Michael H Gold, MD, FAAD, in a release. “It was one of the first cosmeceutical brands I brought into my office more than 30 years ago, and today it remains a name my patients recognize and trust. That level of brand equity makes integration seamless. The Pro Pen reflects that same standard—thoughtfully designed, easy to use, and able to deliver the precise depths needed for consistent, results-driven microneedling treatments.”
Photo caption: PRO Pen
Photo credit: Dermalogica