
Reconstructing the soft tissue of a mangled upper extremity presents unique challenges to the plastic surgeon.
The successful reconstruction of these complex defects requires a thorough knowledge of extremity anatomy and function, as well as proficiency with a wide array of surgical techniques. Functional and aesthetic concerns must be addressed so as to optimize the outcome, and no single surgical approach is sufficient for all defects.
From a functional standpoint, soft-tissue coverage should provide protection for the neurovascular, musculoskeletal, and articular aspects of the hand, and allow unrestricted range of motion. Ideally, the soft-tissue envelope should be sensate, durable, and able to withstand routine daily trauma. Also, because the hand and upper extremity are highly visible during social interactions, efforts should be made to provide an aesthetically acceptable result.
DEALING WITH TRAUMA
When a surgeon is confronted with a devastating upper-extremity injury, the initial phase of management should focus on assessment of the injury. This “inventory” considers the anatomic and functional nature of the defect. Often, a simple chart that indicates the injured structures is helpful to prioritize the management and to determine the reconstructive requirements.
Based on the nature and extent of the injury, a surgical plan is then formulated. For many high-impact industrial and farm injuries, this may require a staged approach. For heavily contaminated wounds, thorough cleansing and debridement take precedence over soft-tissue coverage. Removing foreign bodies and debriding devitalized and infected tissue are essential prior to wound closure. Occasionally, serial debridement at 48- to 72-hour intervals is required to properly assess tissue viability.
Accurate assessment of tissue viability depends upon the extent of debridement. Leaving unperfused or heavily colonized tissue buried beneath soft-tissue flaps will likely result in a subsequent wound infection. During the interval between debridement and soft-tissue coverage, negative-pressure dressings may be helpful by decreasing the frequency of dressing changes and preventing the desiccation of critical structures.
After bacteriologic control of the wound is established, the second phase of management is directed at soft-tissue coverage. However, the surgical management of these injuries is just the beginning of the rehabilitation process. Maximum functional return is achieved only with a multidisciplinary approach that involves the patient, surgeon, and hand therapist.
For small, isolated injuries, local-tissue rearrangement, such as a Z-plasty, is useful. These adjacent tissue transfers are commonly used to deepen web spaces or release contractures in order to facilitate pinch and grasp functions. However, because major trauma, such as industrial crush injuries, may disrupt blood flow to a significant portion of the extremity, local or regional flaps should be used cautiously. These flap options may be located within the injury zone and may not provide reliable coverage.
In addition, surgical incisions in the affected hand and wrist must be well planned so as to avoid scar contractures that may interfere with the form or function of the extremity. Therefore, when addressing defects of larger size and greater complexity, additional coverage options should be considered. Quite frequently, these composite defects require a combination of skin grafts and flaps.
GRAFT CHOICES
When considering a skin graft, the choice between split-thickness and full-thickness grafts depends on several factors. The advantages of split-thickness grafts include the abundance of donor sites, the improved likelihood of graft take, and the ease of contouring the graft over irregular surfaces. However, split-thickness grafts are predisposed toward contracture and pigment discrepancy when compared with native skin.
Conversely, full-thickness grafts are less likely to contract and are typically more aesthetically pleasing. In addition, full-thickness grafts are likely to be more durable than their split-thickness counterparts. However, there is a paucity of donor sites on the body for full-thickness grafts, and these grafts may be more difficult to contour over complex 3D topography.
Another skin-graft donor-site option is glabrous skin, such as the plantar foot. Glabrous skin grafts provide skin that more closely approximates the color and texture of the glabrous skin of the palm. These grafts are useful for coverage of small, volar hand defects, especially in dark-pigmented individuals.
To extend the use of full-thickness skin grafts, nontraditional donor sites should be considered. Although skin is commonly harvested from the groin, abundant tissue can be provided from sites such as the lower abdomen. In fact, coverage of even large upper-extremity wounds can be successfully closed with full-thickness grafts harvested using an “abdominoplasty approach.” An additional benefit of this approach is an improvement in the appearance of the donor site. This transforms potential donor-site liability, such as scarring, into the benefit of improved abdominal contour.
Other skin-graft considerations include the choice between meshed versus sheet grafts. In certain situations, such as major burns, donor skin is limited. In these cases, meshed grafts are required for increased surface-area coverage. When donor skin is sufficient, nonmeshed sheet grafts provide improved cosmesis. This may allow patients to wear short-sleeved shirts with minimal scar stigma.
Finally, negative-pressure dressings can be applied to stabilize skin grafts during the postoperative period. Negative pressure is useful in providing a uniform, stabilizing force for skin grafts. This is especially helpful for grafts located over joints or irregular surfaces. In addition, negative-pressure dressings may help minimize shear on these mobile surfaces.
The typical skin-graft dressing includes a nonadhering gauze applied directly over the graft, followed by a polyurethane foam sponge. This dressing is left intact for 4 days, and negative pressure is applied continuously at 125 mm Hg. On the fourth postoperative day, the negative-pressure dressing is removed and a graded compression dressing is applied.
FLAPS MAY BE BETTER
Although skin grafts may be the least complicated option for wound closure, they do not necessarily provide the optimal functional or aesthetic result. For example, a skin graft placed over peritendon or periosteum may lead to impaired tendon gliding or restricted joint function. To this end, a variety of soft-tissue flaps can be used.
Flap reconstruction has the advantage of importing well-vascularized tissue into the wound bed. Also, flap reconstruction may be required when other procedures, such as tendon grafts, bone grafts, or internal fixation, are performed simultaneously.
Local flaps are typically reserved for smaller defects; these transposition, advancement, rotational, or local perforator flaps can provide thin, sensate soft-tissue coverage. However, for more extensive defects, local flaps do not provide adequate quantities of soft tissue. Furthermore, in extensive crush injuries, the injury zone may extend beyond the overtly necrotic tissue into the adjacent, surrounding local and regional tissue. Care must be taken when designing local flaps to remain outside the injury zone.
If local flaps are judged to be inadequate, regional flaps may provide a reasonable alternative. These tissue transfers may require skin-graft coverage of the donor-site defect, as well as a secondary procedure to divide and inset the flap. The neurovascular-island flap and cross-finger flap are typical regional flaps; their usefulness, as with local flaps, is limited to relatively small defects. In addition, the potential for joint stiffness at the donor site may limit the widespread use of these flaps. In general, they are reserved for small defects in younger patients.
The fillet flap remains an important option when an adjacent digit cannot be salvaged. This flap, which consists of soft tissue from an otherwise unsalvageable digit, can be stripped of its bony and tendonous elements and used for adjacent soft-tissue coverage. Additional regional flap options that provide more abundant soft tissue include the reverse radial forearm flap and the reverse posterior interosseous flap.
The reverse radial forearm flap relies on retrograde flow through the radial artery following distal artery division. Venous drainage is provided by the venae comitantes. This fasciocutaneous flap can be rotated 180° on its proximal pedicle and provides reliable, distal soft-tissue coverage. The donor site is typically covered with a full-thickness or split-thickness skin graft. Confirmation of a patent palmar arch is mandated prior to use. This flap may be especially useful in patients whose health status precludes a lengthy microvascular reconstruction. However, using this flap does require division of a major artery in the affected, traumatized extremity.
The reverse posterior interosseous flap may also be used for soft-tissue coverage of the dorsal wrist and hand. Once again, care must be taken when using this flap in a traumatized extremity. Injury to the anterior interosseous artery precludes the use of this reverse flap.
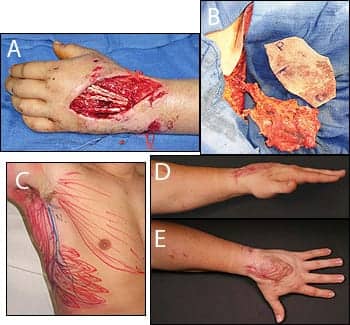 |
| Figure 1. A left hand dorsal degloving injury and extensor tendon rupture (A). Preoperative outline of serratus fascial flap (B). Serratus fascial flap harvested with adjacent template of defect (C). Two postoperative views (D and E). PHOTOS COURTESY OF LOREN S. SCHECHTER, MD |
DISTANT FLAPS
Because of the size limitation of local and regional flaps and the potential involvement of these flaps in the injury zone, distant flaps may be required in cases of extensive tissue loss. An additional advantage of these flaps is the option for composite tissue transfer. These flaps may permit the transfer of bone, tendon, nerve, or functional muscles. Although most commonly transferred using microsurgical techniques, distant flaps may remain attached to their donor site, most commonly at the trunk.
The groin flap has traditionally been the favored pedicle flap for upper-extremity reconstruction. This flap, based on the superficial circumflex iliac vessels, is relatively straightforward to elevate, and is capable of providing significant amounts of reliable skin cover. The groin flap is most commonly used to cover distal, dorsal finger, hand, and forearm wounds. It is typically bulky upon initial inset and requires secondary procedures to thin the flap and improve its contour. The groin flap can be split distally to cover multiple discrete surfaces, if necessary.
Other pedicle flaps, such as the thoracoacromial flap, have been used to cover moderate-sized wounds of the dorsal hand. These pedicle flaps necessitate prolonged extremity immobilization, require pedicle division and inset as a secondary surgical procedure, often lack sensibility, and may be excessively bulky. In addition, the donor site may leave an unsightly defect. Although these factors may limit the utility of these flaps, they still play a role in the plastic surgeon’s armamentarium when options for coverage are limited.
MICROSURGICAL RECONSTRUCTION
Microsurgical flap reconstruction of large, complex extremity wounds offers many advantages over other reconstructive techniques. The reconstruction can often be performed during a single operation. This may reduce the need for prolonged immobilization of the extremity and secondary procedures to divide and inset the flap.
There are many well-described flaps for microsurgical reconstruction of the upper extremity. Flap choice depends on the reconstructive requirements and the donor-site options. Flaps such as the latissimus dorsi, serratus anterior, and gracilis may provide soft-tissue coverage as well as functional muscles. Other flaps, such as the lateral arm flap, may provide sensate soft-tissue coverage.
For small- to medium-sized defects, thin soft-tissue coverage may be provided with fascial flaps covered with skin grafts (Figure 1). Additionally, the fibula flap can provide vascularized bone and soft-tissue coverage for reconstructing composite defects of the extremity.
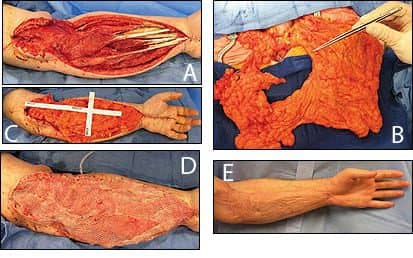 |
| Figure 2. An open wound of left upper extremity following debridement for necrotizing soft tissue infection (A). The harvest and lengthening of omental free flap (B), the omental free flap inset (C). Immediate skin graft applied (D) and a postoperative view (E). |
In addition, in cases of massive tissue loss, microsurgical reconstruction allows the transfer of large amounts of vascularized, pliable soft tissue in a single stage. Of special interest in extensive upper-extremity defects is the omental free flap.
The omental free flap has a long and reliable vascular that can be contoured to cover large 3D defects, provide a gliding surface for tendons, and has additional angiogenic and immunologic capacities. Additionally, laparoscopic assisted harvest can minimize donor site morbidity (Figure 2).
Each of these flaps has unique characteristics that offer multiple options for functional reconstruction of composite defects.
Although microsurgical flap recon-struction necessitates longer surgeries by minimizing the need for secondary procedures for flap division, inset, and debulking, and permitting earlier mobilization, this complex reconstructive option may shorten hospital stay and permit faster rehabilitation.
A relatively recent adjunct to upper-extremity repair is the use of tissue expanders. Although tissue expanders are not commonly used in the initial management of these wounds, they may be useful for secondary reconstruction.
By taking advantage of regional skin laxity and the native viscoelastic properties of surrounding skin, adjacent skin can be slowly stretched to provide increased surface area for resurfacing prior skin grafts, areas of contracture, or aesthetically displeasing scars. Multiple expanders can be used simultaneously, and skin can be repositioned and re-expanded when necessary.
CONCLUSION
The successful reconstruction of complex upper-extremity wounds remains a challenge to plastic surgeons. The goals include optimizing the function and appearance of the extremity.
The surgeon who can draw from the variety of available reconstructive techniques will be able to provide the patient with an extremity that permits maximal recovery of use. However, comprehensive rehabilitation requires a commitment from the patient, surgeon, and therapist.
This multidisciplinary approach helps maximize the patient’s physical and psychological recuperation, as well as his or her reintegration into society.
Loren S. Schechter, MD, is in private practice in Morton Grove, Ill, and serves as chief of plastic surgery at Chicago Medical School of Rosalind Franklin University and division director of plastic surgery at Lutheran General Hospital in Park Ridge and St Francis Hospital in Evanston, Ill. He is certified by the American Board of Plastic Surgery and can be reached at (847) 967-5122 or .





